Медикамент запрещен для реализации в аптеках.
|
||
|
||
|
||
|
||
|
||
|
||
|
||
|
||
|
||
|

Информация о лекарственных препаратах, размещенная на AptekaMos.ru, не должна использоваться неспециалистами для самостоятельного принятия решения об их покупке и применении без консультации врача.
Свидетельство о регистрации средства массовой информации ЭЛ № ФС77-44705 выдано Федеральной службой по надзору в сфере связи, информационных технологий и массовых коммуникаций (Роскомнадзор) 21 апреля 2011 года.
Package insert / product label
Generic name: albuterol sulfate
Dosage form: inhalation solution
Drug class: Adrenergic bronchodilators
Albuterol Inhalation Solution Description
Albuterol sulfate inhalation solution is a sterile, clear, colorless solution of the sulfate salt of racemic albuterol, albuterol sulfate. Albuterol sulfate is a relatively selective beta
2-adrenergic bronchodilator (see
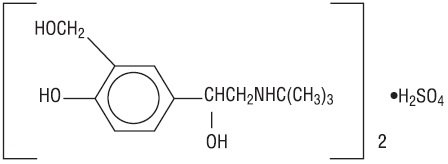
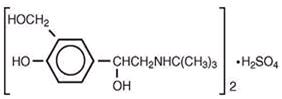
CLINICAL PHARMACOLOGY). The chemical name for albuterol sulfate is α
1 [(tert-butylamino) methyl]-4-hydroxy-m-xylene-α,α’-diol sulfate (2:1) (salt), and its established chemical structure is as follows:
The molecular weight of albuterol sulfate is 576.7 and the molecular formula is (C
13H
21NO
3)
2 • H
2SO
4. Albuterol sulfate USP is a white or practically white powder, freely soluble in water, slightly soluble in alcohol, in ether and practically insoluble in chloroform. The World Health Organization recommended name for albuterol is salbutamol.
Albuterol sulfate inhalation solution is supplied in two strengths in unit dose vials. Each unit dose vial contains either 0.63 mg of albuterol equivalent to 0.75 mg of albuterol sulfate or 1.25 mg of albuterol equivalent to 1.5 mg of albuterol sulfate with sodium chloride and sulfuric acid in a 3-mL isotonic, sterile, aqueous solution. Sodium chloride is added to adjust isotonicity of the solution and sulfuric acid is added to adjust pH of the solution to 3.5 (see
HOW SUPPLIED).
Albuterol sulfate inhalation solution does not require dilution prior to administration by nebulization. For albuterol sulfate inhalation solution, like all other nebulized treatments, the amount delivered to the lungs will depend on patient factors, the jet nebulizer utilized, and compressor performance. Using the Pari LC Plus
TM nebulizer (with face mask or mouthpiece) connected to a Pari PRONEB
TM compressor, under in vitro conditions, the mean delivered dose from the mouth piece (% nominal dose) was approximately 43% of albuterol (1.25 mg strength) and 39% of albuterol (0.63 mg strength) at a mean flow rate of 3.6 L/min. The mean nebulization time was 15 minutes or less. Albuterol sulfate inhalation solution should be administered from a jet nebulizer at an adequate flow rate, via a mouthpiece or face mask (see
DOSAGE AND ADMINISTRATION).
Albuterol Inhalation Solution — Clinical Pharmacology
The prime action of beta-adrenergic drugs is to stimulate adenyl cyclase, the enzyme which catalyzes the formation of cyclic-3′,5′-adenosine monophosphate (cyclic AMP) from adenosine triphosphate (ATP). The cyclic AMP thus formed mediates the cellular responses. In vitro studies and in vivo pharmacologic studies have demonstrated that albuterol has a preferential effect on beta
2-adrenergic receptors compared with isoproterenol. While it is recognized that beta
2-adrenergic receptors are the predominant receptors in bronchial smooth muscle, recent data indicate that 10% to 50% of the beta- receptors in the human heart may be beta
2-receptors. The precise function of these receptors, however, is not yet established. Controlled clinical studies and other clinical experience have shown that inhaled albuterol, like other beta-adrenergic agonist drugs, can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or electrocardiographic changes. Albuterol is longer acting than isoproterenol in most patients by any route of administration because it is not a substrate for the cellular uptake processes for catecholamines nor for catechol-O-methyl transferase.
Pharmacokinetics: Studies in asthmatic patients have shown that less than 20% of a single albuterol dose was absorbed following either intermittent positive-pressure breathing (IPPB) or nebulizer administration; the remaining amount was recovered from the nebulizer and apparatus, and expired air. Most of the absorbed dose was recovered in urine collected during the 24 hours after drug administration. Following oral administration of 4 mg albuterol, the elimination half-life was five to six hours. Following a 3 mg dose of nebulized albuterol in adults, the mean maximum albuterol plasma level at 0.5 hours was 2.1 ng/mL (range, 1.4 to 3.2 ng/mL). The pharmacokinetics of albuterol following administration of 0.63 mg or 1.25 mg albuterol sulfate inhalation solution by nebulization have not been determined in children 2 to 12 years old.
Animal Pharmacology/Toxicology: Intravenous studies in rats with albuterol sulfate have demonstrated that albuterol crosses the blood-brain barrier and reaches brain concentrations amounting to approximately 5% of plasma concentrations. In structures outside the blood-brain barrier (pineal and pituitary glands), albuterol concentrations were found to be 100 times those found in whole brain.
Studies in laboratory animals (minipigs, rodents, and dogs) have demonstrated the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta-agonists and methylxanthines are administered concurrently. The clinical significance of these findings is unknown.
Clinical Trials: The safety and efficacy of albuterol sulfate inhalation solution was evaluated in a 4-week, multi-center, randomized, double-blind, placebo-controlled, parallel group study in 349 children 6 to 12 years of age with mild-to-moderate asthma (mean baseline FEV
1 60% to 70% of predicted). Approximately half of the patients were also receiving inhaled corticosteroids. Patients were randomized to receive albuterol sulfate inhalation solution 0.63 mg, albuterol sulfate inhalation solution 1.25 mg, or placebo three times a day administered via a Pari LC Plus
TM nebulizer and a Pari PRONEB
TM compressor. Racemic albuterol, delivered by a chlorofluorocarbon (CFC) metered dose inhaler (MDI) or nebulized, was used on an as-needed basis as the rescue medication.
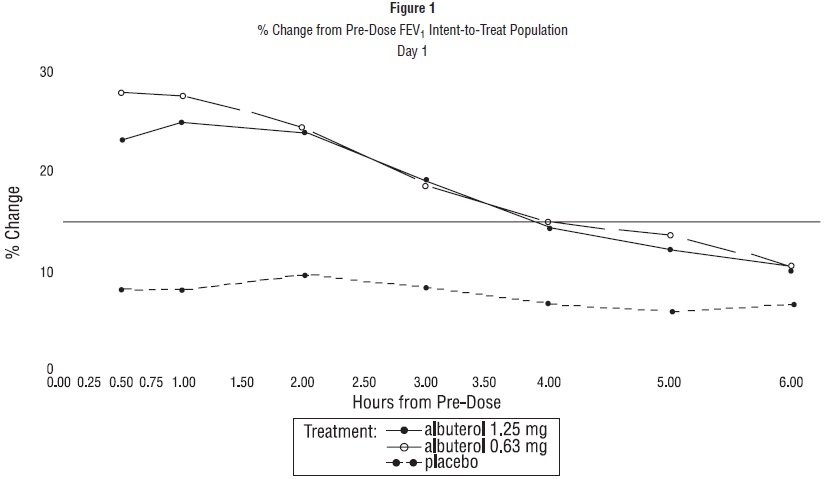
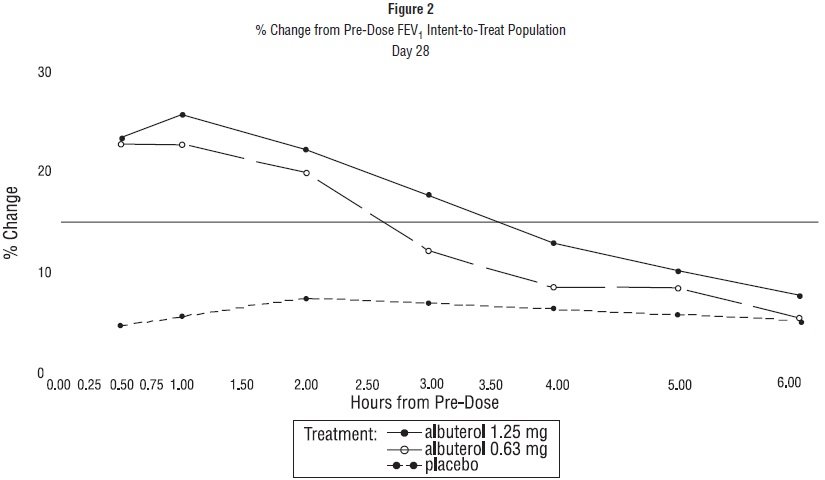
Efficacy, as measured by the mean percent change from baseline in the area under the 6-hour curve for FEV
1, was demonstrated for both active treatment regimens (n=112 [1.25 mg group] and n=110 [0.63 mg group]) compared with placebo (n=110) on day 1 and day 28. Figures 1 and 2 illustrate the mean percentage change from pre-dose FEV
1 on day 1 and day 28, respectively. The mean baseline FEV
1 for all patients was 1.49 L.
The onset of a 15% increase in FEV
1 over baseline for both doses of albuterol sulfate inhalation solution was seen at 30 minutes (the first post-dose assessment). The mean time to peak effect was approximately 30 to 60 minutes for both doses on day 1 and after 4 weeks of treatment. The mean duration of effect, as measured by a >15% increase from baseline in FEV
1, was approximately 2.5 hours for both doses on day 1 and approximately 2 hours for both doses after 4 weeks of treatment. In some patients, the duration of effect was as long as 6 hours.
Indications and Usage for Albuterol Inhalation Solution
Albuterol sulfate inhalation solution is indicated for the relief of bronchospasm in patients 2 to 12 years of age with asthma (reversible obstructive airway disease).
Contraindications
Albuterol sulfate inhalation solution is contraindicated in patients with a history of hypersensitivity to any of its components.
Warnings
Paradoxical Bronchospasm: As with other inhaled beta-adrenergic agonists, albuterol sulfate inhalation solution is contraindicated in patients with a history of hypersensitivity to any of its components.can produce paradoxical bronchospasm, which may be life threatening. If paradoxical bronchospasm occurs, albuterol sulfate inhalation solution should be discontinued immediately and alternative therapy instituted. It should be noted that paradoxical bronchospasm, when associated with inhaled formulations, frequently occurs with the first use of a new canister or vial.
Use of Anti-Inflammatory Agents: The use of beta-adrenergic bronchodilators alone may not be adequate to control asthma in many patients. Early consideration should be given to adding anti-inflammatory agents (e.g., corticosteroids).
Deterioration of Asthma: Asthma may deteriorate acutely over a period of hours or chronically over several days or longer. If the patient needs more doses of albuterol sulfate inhalation solution than usual, this may be a marker of destabilization of asthma and requires reevaluation of the patient and the treatment regimen, giving special consideration of the possible need for anti-inflammatory treatment (e.g., corticosteroids).
Fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs and with the home use of nebulizers. It is, therefore, essential that the physician instruct the patient in the need for further evaluation, if his/her asthma becomes worse.
Cardiovascular Effects: Albuterol sulfate inhalation solution, like other beta-adrenergic agonists, can produce a clinically significant cardiovascular effect in some patients as measured by pulse rate, blood pressure, and/or symptoms. Although such effects are uncommon for albuterol sulfate inhalation solution at recommended doses, if they occur, the drug may need to be discontinued. In addition, beta-agonists have been reported to produce ECG changes, such as flattening of the T- wave, prolongation of the QTc interval, and ST segment depression. The clinical significance of these findings is unknown. Therefore, albuterol sulfate inhalation solution like all other sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension.
Immediate Hypersensitivity Reactions: Immediate hypersensitivity reactions may occur after administration of albuterol as demonstrated by rare cases of urticaria, angioedema, rash, bronchospasm, and oropharyngeal edema.
Precautions
General
Large doses of intravenous albuterol have been reported to aggravate pre-existing diabetes mellitus and ketoacidosis. As with other beta-agonists, inhaled and intravenous albuterol may produce a significant hypokalemia in some patients, possibly through intracellular shunting, which has the potential to produce adverse cardiovascular effects. The decrease is usually transient, not requiring potassium supplementation.
Information for Patients
The action of albuterol sulfate inhalation solution may last up to six hours, and therefore it should not be used more frequently than recommended. Do not increase the dose or frequency of medication without consulting your physician. If you find that treatment with albuterol sulfate inhalation solution becomes less effective for symptomatic relief, your symptoms become worse, and/or you need to use the product more frequently than usual, you should seek medical attention immediately. All asthma medication should only be used under the supervision and direction of a physician. Common effects with medications such as albuterol sulfate inhalation solution include palpitations, chest pain, rapid heart rate, tremor, or nervousness.
If you are pregnant or nursing, contact your physician about the use of albuterol sulfate inhalation solution. Effective and safe use of albuterol sulfate inhalation solution includes an understanding of the way it should be administered.
If the solution in the vial changes color or becomes cloudy, you should not use it.
The drug compatibility (physical and chemical), clinical efficacy, and safety of albuterol sulfate inhalation solution, when mixed with other drugs in a nebulizer, has not been established.
See illustrated Patient’s Instructions for Use.
Drug Interactions
Other short-acting sympathomimetic aerosol bronchodilators or epinephrine should not be used concomitantly with albuterol sulfate inhalation solution.
Albuterol sulfate inhalation solution should be administered with extreme caution to patients being treated with monoamine oxidase inhibitors or tricyclic antidepressants or within 2 weeks of discontinuation of such agents, since the action of albuterol on the vascular system may be potentiated.
Beta-receptor blocking agents not only block the pulmonary effect of beta-agonists, such as albuterol sulfate inhalation solution, but may produce severe bronchospasm in asthmatic patients. Therefore, patients with asthma should not normally be treated with beta-blockers. However, under certain circumstances (e.g., prophylaxis after myocardial infarction), there may be no acceptable alternatives to the use of beta-adrenergic blocking agents in patients with asthma. In this setting, cardioselective beta-blockers should be considered, although they should be administered with caution.
The ECG changes and/or hypokalemia that may result from the administration of non- potassium sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, especially when the dose of the beta-agonist is exceeded. Although the clinical significance of these effects is unknown, caution is advised in the co-administration of beta-agonists with non-potassium sparing diuretics.
Mean decreases of 16% to 22% in serum digoxin levels were demonstrated after single dose intravenous and oral administration of albuterol, respectively, to normal volunteers who had received digoxin for 10 days. The clinical significance of these findings for patients with obstructive airway disease who are receiving albuterol and digoxin on a chronic basis is unclear. Nevertheless, it would be prudent to carefully evaluate the serum digoxin levels in patients who are currently receiving digoxin and albuterol.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
In a 2-year study in Sprague-Dawley rats, albuterol sulfate caused a significant dose-related increase in the incidence of benign leiomyomas of the mesovarium and above dietary doses of 2 mg/kg (approximately equivalent to the maximum recommended daily inhalation dose for albuterol sulfate inhalation solution on a mg/m
2 basis). In another study, this effect was blocked by the co-administration of propranolol, a non-selective beta-adrenergic antagonist.
In an 18-month study in CD-1 mice, albuterol sulfate showed no evidence of tumorigenicity at dietary doses up to 500 mg/kg (approximately 140 times the maximum recommended daily inhalation dose of albuterol sulfate inhalation solution on a mg/m
2 basis). In a 22-month study in Golden hamsters, albuterol sulfate showed no evidence of tumorigenicity at dietary doses up to 50 mg/kg (approximately 20 times the maximum recommended daily inhalation dose of albuterol sulfate inhalation solution on a mg/m
2 basis).
Albuterol sulfate was not mutagenic in the Ames test or a mutation test in yeast. Albuterol sulfate was not clastogenic in a human peripheral lymphocyte assay or in an AH
1 strain mouse micronucleus assay.
Reproduction studies in rats demonstrated no evidence of impaired fertility at oral doses of albuterol sulfate up to 50 mg/kg (approximately 30 times the maximum recommended daily inhalation dose of albuterol sulfate inhalation solution on a mg/m
2 basis).
Pregnancy
Teratogenic Effects: Albuterol has been shown to be teratogenic in mice. A study in CD-1 mice given albuterol subcutaneously showed cleft palate formation in 5 of 111 (4.5%) fetuses at 0.25 mg/kg (less than the maximum recommended daily inhalation dose of albuterol sulfate inhalation solution on a mg/m
2 basis) and cleft palate formation in 10 of 108 (9.3%) fetuses at 2.5 mg/kg (approximately equal to the maximum recommended daily inhalation dose of albuterol sulfate inhalation solution on a mg/m
2 basis). The drug did not induce cleft palate formation when administered subcutaneously at a dose of 0.025 mg/kg (less than the maximum recommended daily inhalation dose of albuterol sulfate inhalation solution on a mg/m
2 basis). Cleft palate formation also occurred in 23 of 72 (30.5%) fetuses from females treated subcutaneously with 2.5 mg/kg isoproterenol (positive control). A reproduction study in Stride rabbits revealed cranioschisis in 7 of 19 (37%) fetuses when albuterol sulfate was administered orally at 50 mg/kg (approximately 60 times the maximum recommended daily inhalation dose of albuterol sulfate inhalation solution on a mg/m
2 basis).
A study in which pregnant rats were dosed with radiolabeled albuterol sulfate demonstrated that drug-related material was transferred from the maternal circulation to the fetus.
There are no adequate and well-controlled studies of the use of albuterol sulfate in pregnant women. Albuterol should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
During worldwide marketing experience, various congenital anomalies, including cleft palate and limb defects, have been reported in the offspring of patients being treated with albuterol. Some of the mothers were taking multiple medications during their pregnancies. Because no consistent pattern of defects can be discerned, a relationship between albuterol use and congenital anomalies has not been established.
Labor and Delivery
Oral albuterol has been shown to delay pre-term labor in some reports. There are presently no well-controlled studies that demonstrate that it will stop pre-term labor or prevent labor at term. Because of the potential for beta agonist interference with uterine contractility, use of albuterol sulfate inhalation solution for relief of bronchospasm during labor should be restricted to those patients in whom the benefits clearly outweigh the risk.
Albuterol has not been approved for the management of pre-term labor. The benefit:risk ratio when albuterol is administered for tocolysis has not been established. Serious adverse reactions, including pulmonary edema, have been reported following administration of albuterol to women in labor.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because of the potential for tumorigenicity shown for albuterol in some animal studies, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Safety and effectiveness of albuterol sulfate inhalation solution 1.25 mg and 0.63 mg have been established in pediatric patients between the ages of 2 and 12 years. The use of albuterol sulfate inhalation solution in these age groups is supported by evidence from adequate and well-controlled studies of albuterol sulfate inhalation solution in children age 6 to 12 years and published reports of albuterol sulfate trials in pediatric patients 3 years of age and older. The safety and effectiveness of albuterol sulfate inhalation solution in children below 2 years of age have not been established.
Adverse Reactions/Side Effects
Clinical Trial Experience: Adverse events reported in >1% of patients receiving albuterol sulfate inhalation solution and more frequently than in patients receiving placebo in a four-week double- blind study are listed in the following table.
| 1.25 mg Albuterol Sulfate Inhalation Solution (N=115) |
0.63 mg Albuterol Sulfate Inhalation Solution (N=117) |
Placebo
(N=117) |
|
| Asthma Exacerbation |
13 |
11.1 |
8.5 |
| Otitis Media |
4.3 |
0.9 |
0 |
| Allergic Reaction |
0.9 |
3.4 |
1.7 |
| Gastroenteritis |
0.9 |
3.4 |
0.9 |
| Cold Symptoms |
0 |
3.4 |
1.7 |
| Flu Syndrome |
2.6 |
2.6 |
1.7 |
| Lymphadenopathy |
2.6 |
0.9 |
1.7 |
| Skin/Appendage Infection |
1.7 |
0 |
0 |
| Urticaria |
1.7 |
0.9 |
0 |
| Migraine |
0.9 |
1.7 |
0 |
| Chest Pain |
0.9 |
1.7 |
0 |
| Bronchitis |
0.9 |
1.7 |
0.9 |
| Nausea |
1.7 |
0.9 |
0.9 |
There was one case of ST segment depression in the 1.25 mg albuterol sulfate inhalation solution treatment group.
No clinically relevant laboratory abnormalities related to albuterol sulfate inhalation solution administration were seen in this study.
Postmarketing Experience : Metabolic acidosis has been reported after the use of albuterol sulfate inhalation solution. Because this reaction is reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate its frequency or establish a causal relationship to drug exposure.
Overdosage
The expected symptoms with overdosage are those of excessive beta-adrenergic stimulation and/or occurrence or exaggeration of symptoms such as seizures, angina, hypertension or hypotension, tachycardia with rates up to 200 beats per minute, arrhythmias, nervousness, headache, tremor, dry mouth, palpitation, nausea, dizziness, fatigue, malaise, insomnia, and exaggeration of the pharmacological effects listed in ADVERSE REACTIONS. Hypokalemia may also occur. As with all sympathomimetic aerosol medications, cardiac arrest and even death may be associated with abuse of albuterol sulfate inhalation solution. Treatment consists of discontinuation of albuterol sulfate inhalation solution together with appropriate symptomatic therapy. The judicious use of a cardioselective beta-receptor blocker may be considered, bearing in mind that such medication can produce bronchospasm. There is insufficient evidence to determine if dialysis is beneficial for overdosage of albuterol sulfate inhalation solution.
The oral median lethal dose of albuterol sulfate in mice is greater than 2,000 mg/kg (approximately 580 times the maximum recommended daily inhalation dose of albuterol sulfate inhalation solution on a mg/m
2 basis). The subcutaneous median lethal dose of albuterol sulfate in mature rats and small young rats is approximately 450 mg/kg and 2,000 mg/kg, respectively (approximately 260 and 1,200 times the maximum recommended daily inhalation dose of albuterol sulfate inhalation solution on a mg/m
2 basis). The inhalation median lethal dose has not been determined in animals.
Albuterol Inhalation Solution Dosage and Administration
The usual starting dosage for patients 2 to 12 years of age is 1.25 mg or 0.63 mg of albuterol sulfate inhalation solution administered 3 or 4 times daily, as needed, by nebulization. More frequent administration is not recommended.
To administer 1.25 mg or 0.63 mg of albuterol, use the entire contents of one unit-dose vial (3 mL of 1.25 mg or 0.63 mg inhalation solution) by nebulization. Adjust nebulizer flow rate to deliver albuterol sulfate inhalation solution over 5 to 15 minutes.
The use of albuterol sulfate inhalation solution can be continued as medically indicated to control recurring bouts of bronchospasm. During this time most patients gain optimum benefit from regular use of the inhalation solution.
Patients 6 to 12 years of age with more severe asthma (baseline FEV
1 less than 60% predicted), weight >40 kg, or patients 11 to 12 years of age may achieve a better initial response with the 1.25 mg dose.
Albuterol sulfate inhalation solution has not been studied in the setting of acute attacks of bronchospasm. A 2.5 mg dose of albuterol provided by a higher concentration product (2.5 mg albuterol per 3 mL) may be more appropriate for treating acute exacerbations, particularly in children 6 years old and above.
If a previously effective dosage regimen fails to provide the usual relief, medical advice should be sought immediately, as this is often a sign of seriously worsening asthma which would require reassessment of therapy.
The drug compatibility (physical and chemical), clinical efficacy and safety of albuterol sulfate inhalation solution, when mixed with other drugs in a nebulizer have not been established.
The safety and efficacy of albuterol sulfate inhalation solution have been established in clinical trials when administered using the Pari LC Plus
TM nebulizer and Pari PRONEB
TM compressor. The safety and efficacy of albuterol sulfate inhalation solution when administered with other nebulizer systems have not been established.
Albuterol sulfate inhalation solution should be administered via jet nebulizer connected to an air compressor with adequate air flow, equipped with a mouthpiece or suitable face mask.
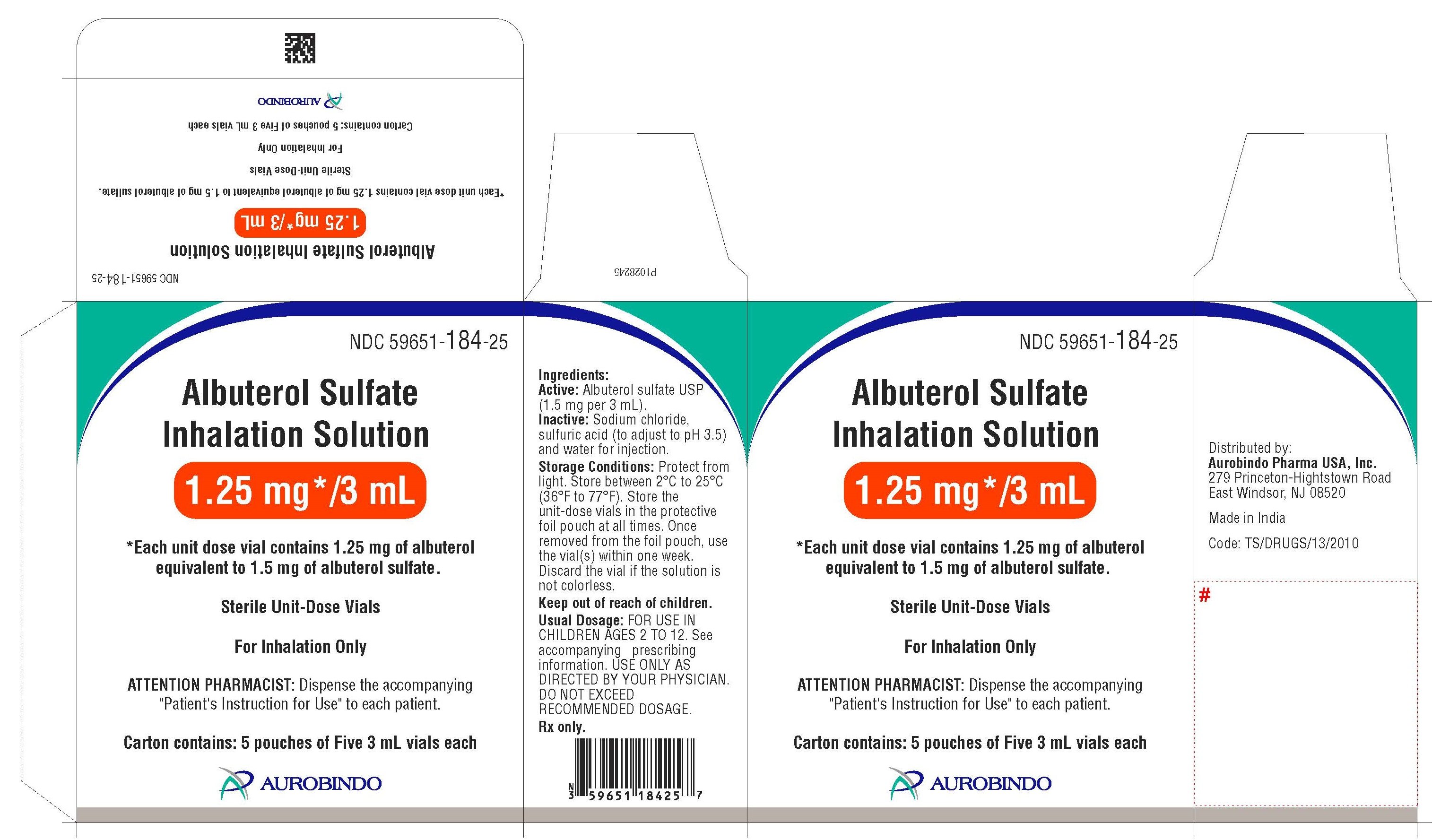
How is Albuterol Inhalation Solution supplied
Albuterol Sulfate Inhalation Solution is supplied as a 3 mL, clear, colorless, sterile, preservative-free, aqueous solution in two different strengths, 0.63 mg/3 mL and 1.25 mg/3 mL, of albuterol (equivalent to 0.75 mg of albuterol sulfate or 1.5 mg of albuterol sulfate per 3 mL) in unit-dose low-density polyethylene (LDPE) vials. Each unit-dose LDPE vial is protected in a foil pouch, and each foil pouch contains 5 unit-dose LDPE vials. Each strength of albuterol sulfate inhalation solution is available in a shelf carton containing multiple foil pouches.
Albuterol Sulfate Inhalation Solution, 0.63 mg/3 mL (potency expressed as albuterol) contains 0.75 mg albuterol sulfate per 3 mL:
Foil pouch of five 3 mL unit-dose vials NDC 59651-183-05
5 such foil pouches in a carton (25 unit-dose vials) NDC 59651-183-25
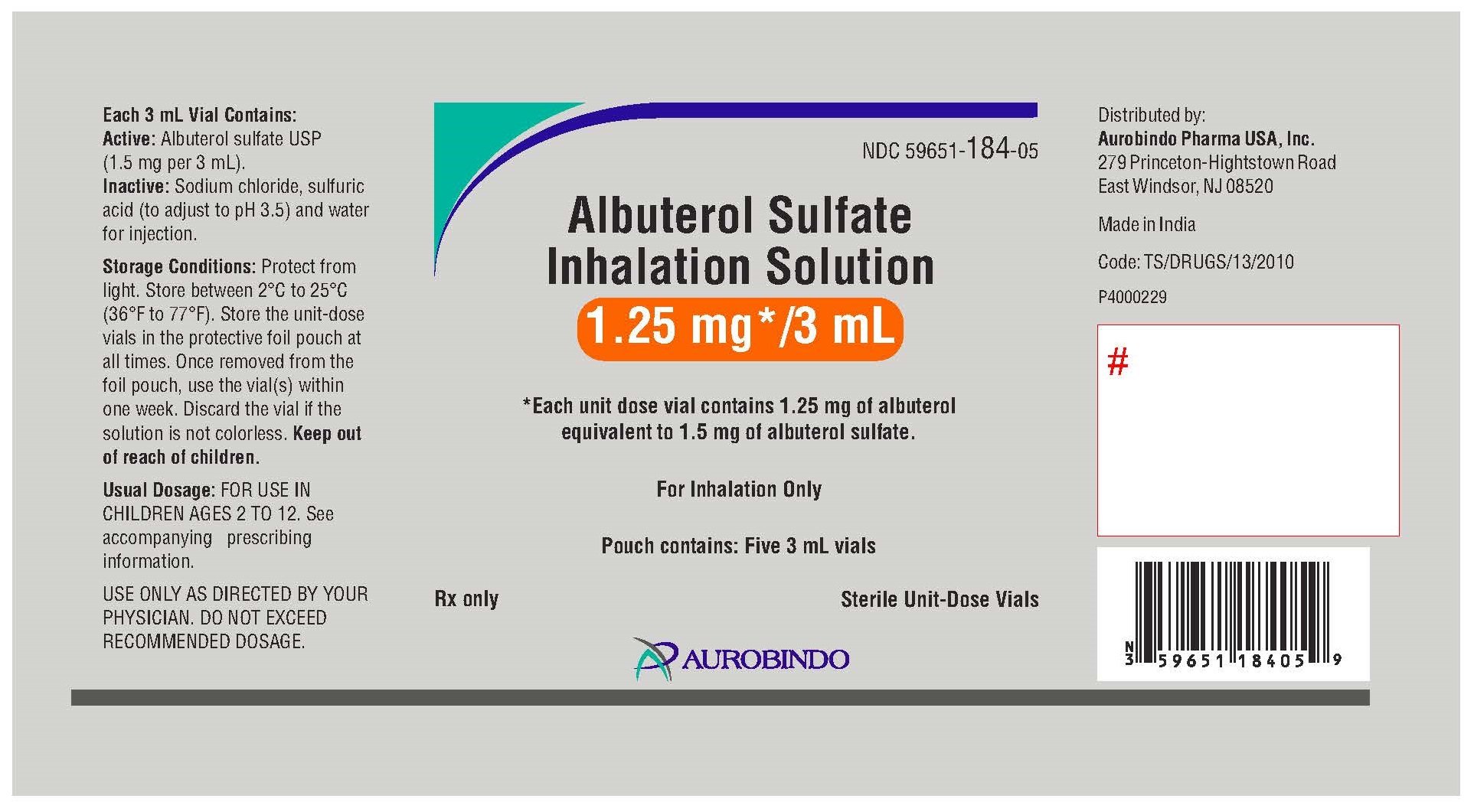
Albuterol Sulfate Inhalation Solution, 1.25 mg/3 mL (potency expressed as albuterol) contains 1.50 mg albuterol sulfate per 3 mL:
Foil pouch of five 3 mL unit-dose vials NDC 59651-184-05
5 such foil pouches in a carton (25 unit-dose vials) NDC 59651-184-25
STORAGE
Store between 2°C to 25°C (36°F to 77°F). Protect from light and excessive heat.
Store unit-dose vials in protective foil pouch at all times. Once removed from the foil pouch, use vial(s) within one week. Discard the vial if the solution is not colorless.
Keep out of the reach of children.
All brands listed are the trademarks of their respective owners and are not trademarks of Luoxin Aurovitas Pharma (Chengdu) Co., Ltd.
Distributed by:
Aurobindo Pharma USA, Inc.
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited, Unit-III,
Plot No: 4, 34 to 48 EPIP,
TSIIC, IDA, Pashamylaram,
Patancheru Mandal, Sangareddy District,
Sangareddy, Telangana — 502307, India.
Revised: August 2022
Patient Information
Albuterol Sulfate Inhalation Solution
(al bue’ ter ol sul’ fate)
0.63 mg*/3 mL and 1.25 mg*/3 mL
(*Equivalent to 0.75 mg of albuterol sulfate or 1.5 mg of albuterol sulfate per 3 mL)
Read the patient information that comes with albuterol sulfate inhalation solution before using it and each time you get a refill for your child. There may be new information. This leaflet does not take the place of talking to your child’s doctor about your child’s medical condition or treatment.
What is albuterol sulfate inhalation solution?
Albuterol sulfate inhalation solution is a medicine that is used for the relief of bronchospasms caused by asthma in children ages 2 to 12 years. Bronchospasm is the tightening and swelling of the muscles around the airways. Albuterol sulfate inhalation solution can help relax these airway muscles for up to 6 hours so that your child may breathe more easily.
Who should not use albuterol sulfate inhalation solution?
Do not give your child albuterol sulfate inhalation solution if he or she is allergic to any of its ingredients. The active ingredient is albuterol sulfate. See the end of this leaflet for a complete list of ingredients.
What should I tell my child’s doctor before giving albuterol sulfate inhalation solution?
Tell your child’s doctor about all of your child’s medical conditions including if your child has:
- Heart problems
- High blood pressure
- Seizures
- A thyroid problem called hyperthyroidism
- Diabetes
Tell your child’s doctor about all the medicines your child takes, including prescription and non-prescription medicines, vitamins and herbal supplements. Albuterol sulfate inhalation solution and some other medicines can affect each other and may cause serious side effects. Especially tell your child’s doctor if your child is taking or using:
- Any short-acting bronchodilator medicines (sometimes called rescue inhalers)
- Epinephrine
- Medicines called monoamine oxidase inhibitors (MAOIs) or tricyclic anti-depressants or has stopped taking them in the past 2 weeks. These medicines are usually used for mental problems.
- Medicines called beta-blockers (used for heart problems and high blood pressure)
- Certain diuretic medicines (water pills)
- Digoxin
Know the medicines your child takes. Keep a list of them and show it to your child’s doctor and pharmacist each time your child gets a new medicine.
How should albuterol sulfate inhalation solution be given?
Read the Patient’s Instructions for Use that comes with albuterol sulfate inhalation solution. Ask your pharmacist for these instructions if they are not with your medicine. Keep the instructions with albuterol sulfate inhalation solution because you may want to read them again.
- Give albuterol sulfate inhalation solution exactly as prescribed for your child. Do not change your child’s dose or how often it is used without talking to your child’s doctor first.
- Albuterol sulfate inhalation solution is breathed into the lungs. Albuterol sulfate inhalation solution is used with a special breathing machine called a nebulizer. Do not mix other medicines with albuterol sulfate inhalation solution in the nebulizer. Do not use albuterol sulfate inhalation solution that is not clear and colorless.
- Call your child’s doctor or get emergency help right away if your child’s breathing is not helped or gets worse during treatment with albuterol sulfate inhalation solution.
- Call your child’s doctor right away if your child needs to use albuterol sulfate inhalation solution more often than prescribed.
- Albuterol sulfate inhalation solution has not been studied for treating acute attacks of bronchospasm (rescue use). Your child may need a different medicine for rescue use.
- If you give your child too much albuterol sulfate inhalation solution, call your child’s doctor right away.
What are the side effects with albuterol sulfate inhalation solution?
Albuterol sulfate inhalation solution may cause the following serious side effects:
- Worsening of the tightening and swelling of the muscles around your child’s airways (bronchospasm). This side effect can be life threatening. Call your child’s doctor or get emergency help right away if your child’s breathing is not helped or gets worse during treatment with albuterol sulfate inhalation solution.
-
Serious and life threatening allergic reactions. Symptoms of a serious allergic reaction include:
- Hives, rash
- Swelling of your child’s face, eyelids, lips, tongue, or throat, and trouble swallowing
- Worsening of your child’s breathing problems such as wheezing, chest tightness or shortness of breath
- Shock (loss of blood pressure and consciousness).
The most common side effects with albuterol sulfate inhalation solution include a fast or irregular heartbeat, chest pain, shakiness, or nervousness.
How should albuterol sulfate inhalation solution be stored?
- Store albuterol sulfate inhalation solution at room temperature, 2°C to 25°C (36°F to 77°F) in its tightly closed container.
- Protect vials from light before use. Therefore, keep unused vials in the foil pouch or carton. Once removed from the foil pouch, use vial(s) within one week.
- Do not use albuterol sulfate inhalation solution after the expiration (EXP) date printed on the vial. Do not use albuterol sulfate inhalation solution that is not clear and colorless.
- Safely, discard albuterol sulfate inhalation solution that is out-of-date or no longer needed.
- Keep albuterol sulfate inhalation solution and all medicines out of the reach of children.
General Information about albuterol sulfate inhalation solution
Medicines are sometimes prescribed for conditions that are not mentioned in the patient information leaflets. Do not use albuterol sulfate inhalation solution for a condition for which it was not prescribed. Do not give albuterol sulfate inhalation solution to other people, even if they have the same symptoms your child has. It may harm them.
This leaflet summarizes the most important information about albuterol sulfate inhalation solution. If you would like more information, talk with your child’s doctor. You can ask your child’s doctor or pharmacist for information about albuterol sulfate inhalation solution that is written for health professionals. You can also call the company that makes albuterol sulfate inhalation solution toll free at 1-866-850-2876.
What are the ingredients in albuterol sulfate inhalation solution?
Active Ingredient: albuterol sulfate
Inactive Ingredients: sodium chloride and sulfuric acid
Rx Only
Distributed by:
Aurobindo Pharma USA, Inc.
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited, Unit-III,
Plot No: 4, 34 to 48 EPIP,
TSIIC, IDA, Pashamylaram,
Patancheru Mandal, Sangareddy District,
Sangareddy, Telangana — 502307, India.
Revised: August 2022
Albuterol Sulfate Inhalation Solution
0.63 mg*/3 mL and 1.25 mg*/3 mL
(*Equivalent to 0.75 mg of albuterol sulfate or 1.5 mg of albuterol sulfate per 3 mL)
PATIENT’S INSTRUCTIONS FOR USE
Read this patient information completely every time your prescription is filled as information may have changed. Keep these instructions with your medication, as you may want to read them again.
Albuterol sulfate inhalation solution should only be used under the direction of a physician. Your physician and pharmacist have more information about albuterol sulfate inhalation solution and the condition for which it has been prescribed. Contact them if you have additional questions.
Storing your Medicine
Store albuterol sulfate inhalation solution between 2° and 25° C (36° and 77° F). Vials should be protected from light before use, therefore, keep unused vials in the foil pouch. Do not use after the expiration (EXP) date printed on the vial.
Dose
Albuterol sulfate inhalation solution is supplied as a single-dose, ready-to-use vial containing 3 mL of solution. No mixing or dilution is needed. Use one new vial with each nebulizer treatment.
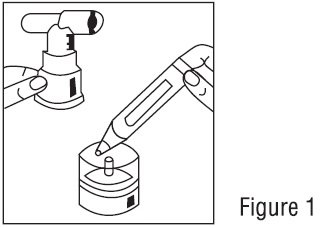
Instructions for Use
1. Remove one vial from the foil pouch. Place remaining vials back into foil pouch for storage.
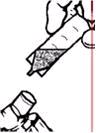
2. Twist the cap completely off the vial and squeeze the contents into the nebulizer reservoir (Figure 1).
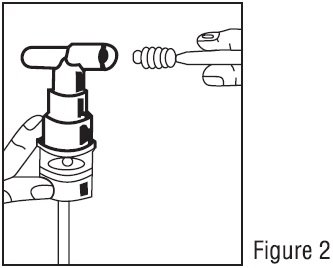
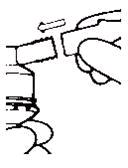
3. Connect the nebulizer to the mouthpiece or face mask (Figure 2).
4. Connect the nebulizer to the compressor.
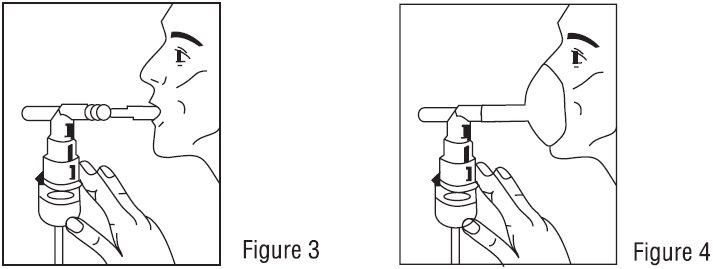
5. Sit in a comfortable, upright position; place the mouthpiece in your mouth (Figure 3) or put on the face mask (Figure 4); and turn on the compressor.
6. Breathe as calmly, deeply and evenly as possible through your mouth until no more mist is formed in the nebulizer chamber (about 5 to 15 minutes). At this point, the treatment is finished.
7. Clean the nebulizer (see manufacturer’s instructions).
Distributed by:
Aurobindo Pharma USA, Inc.
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited, Unit-III,
Plot No: 4, 34 to 48 EPIP,
TSIIC, IDA, Pashamylaram,
Patancheru Mandal, Sangareddy District,
Sangareddy, Telangana — 502307, India.
Revised: August 2022
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL — 0.63 mg per 3 mL — Pouch Label (5 Vials)
NDC 59651-183-05
Albuterol Sulfate
Inhalation Solution
0.63 mg*/3 mL
* Each unit dose vial contains 0.63 mg of
albuterol equivalent to 0.75 mg of albuterol sulfate.
For Inhalation only
Pouch contains: Five 3 mL vials
Rx only Sterile Unit-Dose Vials
AUROBINDO
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL — 0.63 mg per 3 mL – Container-Carton (25 Vials)
NDC 59651-183-25
Albuterol Sulfate
Inhalation Solution
0.63 mg*/3 mL
* Each unit dose vial contains 0.63 mg of albuterol
equivalent to 0.75 mg of albuterol sulfate.
Sterile Unit-Dose Vials
For Inhalation only
ATTENTION PHARMACIST: Dispense the accompanying
“Patient’s Instruction for Use” to each patient.
Carton contains: 5 pouches of Five 3 mL vials each
AUROBINDO
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL – 1.25 mg per 3 mL — Pouch Label (5 Vials)
NDC 59651-184-05
Albuterol Sulfate
Inhalation Solution
1.25 mg*/3 mL
* Each unit dose vial contains 1.25 mg of albuterol
equivalent to 1.5 mg of albuterol sulfate.
For Inhalation only
Pouch contains: Five 3 mL vials
Rx only Sterile Unit-Dose Vials
AUROBINDO
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL — 1.25 mg per 3 mL – Container-Carton (25 Vials)
NDC 59651-184-25
Albuterol Sulfate
Inhalation Solution
1.25 mg*/3 mL
* Each unit dose vial contains 1.25 mg of albuterol
equivalent to 1.5 mg of albuterol sulfate.
Sterile Unit-Dose Vials
For Inhalation only
ATTENTION PHARMACIST: Dispense the accompanying
“Patient’s Instruction for Use” to each patient.
Carton contains: 5 pouches of Five 3 mL vials each
AUROBINDO
Бета-агонисты короткого действия
Альбутерол
ГФА: 90мкг/вдох
Так же, как для взрослых
2 ингаляции каждые 4–6 часов при необходимости и 2 ингаляции за 15–30 минут до физической нагрузки
Альбутерол используется, как средство скорой помощи.
Препарат не рекомендуется для поддерживающего лечения.
Регулярное использование свидетельствует об ухудшении контроля заболевания и необходимости использования дополнительных препаратов.
Дозирующие порошковые ингаляторы имеют такую же эффективность, как и небулайзеры, если пациент умеет пользоваться ингалятором.
Раствор альбутерола для небулайзера можно смешать с другими растворами.
ИСП: 90 мкг/вдох
≥ 4 лет: так же, как и для взрослых
< 4 лет: не используют
2 ингаляции каждые 4–6 часов при необходимости и 2 ингаляции за 15–30 минут до физической нагрузки
Раствор для небулайзера: 5 мг/мл и 0,63, 1,25 и 2,5 мг/3 мл
< 5 лет: 0,63-2,5 мг на 3 мл физиологического раствора каждые 4-6 часов при необходимости
≥ 5 лет: 0,05 мг/кг на 3 мл физиологического раствора каждые 4-6 часов при необходимости (минимум 1,25 мг, максимум 2,5 мг)
1,25-5 мг на 3 мл физиологического раствора каждые 4-6 часов при необходимости
Левалбутерол
ГФА: 45мкг/вдох
< 5 лет: не установлено
≥5 лет: так же, как для взрослых
2 впрыскивания через каждые 4-6 часов при необходимости
Левалбутерол — это R-изомер альбутерола. 0,63 мг соответствует 1,25 мг рацемического альбутерола.
Левалбутерол дает меньше нежелательных явлений.
Раствор для небулайзера: 0,31; 0,63 и 1,25 мг/3 мл и 1,25 мг/0,5 мл
< 5 лет: 0,31-1,25 мг в 3 мл каждые 4 – 6 часов при необходимости
5-11 лет: 0,31-0,63 мг каждые 8 часов при необходимости (максимум 0,63 мг каждые 8 часов)
≥ 12 лет: так же, как для взрослых
0,63-1,25 мг каждые 6-8 часов при необходимости
Бета-агонисты длительного действия (не используются в качестве монотерапии)
Арформотерол
Раствор для ингаляций: 15 мкг/2 мл
Не установлено
15-25 мкг каждые 12 часов
Арфомотерол — это R-изомер формотерола.
Формотерол
Раствор для ингаляций: 20 мкг/2 мл
Не установлено
20 мкг каждые 12 часов
В форме порошкового ингалятора больше не доступен.
Сальметерол
HFA: 21 мкг/впрыскивание (не доступен в США)
≥ 12 лет: так же, как для взрослых
2 вдоха через каждые 12 часов; перед физической нагрузкой следует использовать за 30-60 минут до нее
Продолжительность действия — 12 часов.
При ночной брохиальной астме — одна доза на ночь.
Сальметерол не используется для облегчения симптомов при обострении заболевания.
ИСП: 50 мкг/вдох
< 4 лет: не установлено
≥ 4 лет: так же, как и для взрослых
1 вдох каждые 12 часов и за 30 минут до физической нагрузки
Бета-агонисты ультрадлительного действия (не используются в качестве монотерапии)
Индакатерол
ИСП: 75 мкг/вдох
Не установлено
1 впрыскивание 1 раз в день
—
Олодатерол
Дозированный аэрозольный ингалятор: 2,5 мкг/впрыскивание
Не установлено
2 впрыскивания 1 раз в день
—
Вилантерол
ИСП: 25 мкг/вдох
Не установлено
1 впрыскивание 1 раз в день
Вилантерол доступен только в комбинации с флутиказоном по 100 мкг или 200 мкг.
Антихолинергические препараты
Ипратропий
ГФА: 17 мкг/вдох
< 12 лет: не установлено
≥ 12 лет: так же, как для взрослых
2 вдоха каждые 6 часов при необходимости (максимум 12 вдохов в день)
Ипратропия бромид можно смешивать в одном небулайзере с другими растворами.
Не используется в качестве терапии первой линии.
При долгосрочной поддерживающей терапии регулярное использование не дает явного преимущества, однако его следует добавить к лечению при острых симптомах.
Раствор для небулайзера: 500мкг (0,02%, 2 мл)
< 12 лет: не установлено
≥ 12 лет: так же, как для взрослых
500 мкг каждые 6–8 часов при необходимости
Тиотропия бромид
МИТ: 1,25 мкг/впрыскивание
< 6 лет: не установлено
≥ 6 лет: так же, как для взрослых
2 впырскивания 1 раз в день (максимум 2 впырскивания в день)
Тиотропия бромид имеет более длительный эффект, чем ипратропия бромид.
При астме назначение тиотропия в виде ингаляций с помощью ультрамелкодисперсного ингалятора рекомендуется только в низких дозах.
Порошковый ингалятор: 18мкг/капсула
Не установлено
18 мкг (1 капсула) 1 раз в день
Кортикостероиды (ингаляционные)
Беклометазон
ГФА: 40-80 мкг/вдох
< 4 лет: не установлено
4-11 лет: 1 вдох каждые 12 часов (обычно максимум 80 мкг 2 раза в день)
≥ 12 лет: так же, как для взрослых
1-2 вдоха каждые 12 часов (обычно максимум 320 мкг 2 раза в день)
Выбор дозы зависит от тяжести заболевания и варьирует от 1-2 вдохов до того количества, которое необходимо для контроля заболевания.
При длительном применении возможны системные эффекты.
Максимальная доза — это та доза, при которой происходит угнетение гипоталамо-гипофизарно-надпочечниковой системы.
Если для контроля астмы необходимы более высокие дозы, рекомендуется консультация специалиста.
Будесонид
Порошковый ингалятор: 90 или 180 мкг/впырскивание
< 6 лет: не рекомендуется
≥ 6 лет: начальная доза — 180 мкг 2 раза в день (максимум 360 мкг 2 раза в день)
Начальная доза — 360 мкг 2 раза в день (максимум 720 мкг 2 раза в день)
Раствор для небулайзера: 0,25, 0,5 или 1,0 мг (каждый по 2 мл раствора)
Только для детей от 1 года до 8 лет: если ранее использовались только бронхолитики, начальная доза — 0,5 мг 1 раз в день или 0,25 мг 2 раза в день (максимум 0,5 мг/день)
Если ранее использовались ингаляционные кортикостероиды, начальная доза — 0,5 мг 1 раз в день или 0,25 мг 2 раза в день
Если ранее использовались пероральные кортикостероиды, начальная доза 0,5 мг 2 раза в день или 1 мг 1 раз в день (максимум 1 мг/день)
У взрослых не используется
Циклесонид
ГФА: 80 или 160 мкг/впырскивание
≤ 5 лет: 160 мкг в день
6–11 лет: низкая доза 80 мкг один раз в день, средняя доза > 80–160 мкг один раз в день, высокая доза > 160 мкг один раз в день
≥ 12 лет: так же, как для взрослых
Если ранее применялись только бронхолитики, начальная доза 80 мкг 2 раза в день (максимум 320 мкг 2 раза в день)
Если ранее применялись ингаляционные кортикостероиды, начальная доза 80 мкг 2 раза в день (максимум 640 мкг 2 раза в день)
Если ранее применялись пероральные кортикостероиды, начальная доза 320 мкг 2 раза в день (максимум 640 мкг 2 раза в день)
Флунизолид
ГФА: 80мкг/вдох
< 5 лет: не установлено
5-11 лет: 1 впрыскивание 2 раза в день (максимум 2 впрыскивания 2 раза в день [320 мкг/день])
≥ 12 лет: так же, как для взрослых
2 впырскивания 2 раза в день (максимум 4 впырскивания 2 раза в день [640 мкг/день])
Флутиказона пропионат
ГФА: 44, 110 или 220 мкг/ингаляция
0—4 лет: 50 мкг/день
5–11 лет: обычно общая суточная доза 44–220 мкг.
≥ 12 лет: так же, как для взрослых
Если ранее применялись только бронхолитики, начальная доза 88 мкг 2 раза в день (максимум 440 мкг 2 раза в день)
Если ранее применялись ингаляционные кортикостероиды, начальная доза 88-220 мкг 2 раза в день (максимум 440 мкг 2 раза в день)
Если ранее применялись пероральные кортикостероиды, начальная доза 440-880 мкг 2 раза в день (максимум 880 мкг 2 раза в день)
Порошковый ингалятор: 50, 100 или 250 мкг/впрыскивание
0-4 лет: не установлено
5–11 лет: обычно 50–200 мкг общей суточной дозы (обычно максимум 100 мкг 2 раза в день)
≥ 12 лет: так же, как для взрослых
Если ранее применялись только бронхолитики, начальная доза 100 мкг 2 раза в день (максимум 500 мкг 2 раза в день)
Если ранее применялись ингаляционные кортикостероиды, начальная доза 100-250 мкг 2 раза в день (максимум 500 мкг 2 раза в день)
Если ранее применялись пероральные кортикостероиды, начальная доза 500-1000 мкг 2 раза в день (максимум 1000 мкг 2 раза в день)
Флутиказона фуроат
ИСП: 50, 100 или 200 мкг/впырскивание
0- 4 года: не установлено
5-11 лет: 1 впрыскивание (50 мкг) 1 раз в день.
≥ 12 лет: так же, как для взрослых
Если ранее применялись только бронхолитики, начальная доза 100 мкг 1 раз в день (максимум 200 мкг в день)
Если ранее применялись ингаляционные кортикостероиды, начальная доза 100-200 мкг 1 раз в день (максимум 200 мкг в день)
Мометазон
Порошковый ингалятор: 110 или 220 мкг/впырскивание
< 4 лет: не установлено
4 — 11 лет: 110 мкг 1 раз в день (вечером)
≥ 12 лет: так же, как для взрослых
Если ранее применялись только бронхолитики или ингаляционные кортикостероиды, начальная доза 220 мкг 1 раз в день вечером (максимум 220 мкг 2 раза в день или 440 мкг 1 раз в день вечером)
Если ранее применялись пероральные кортикостероиды, начальная доза 440 мкг 2 раза в день (максимум 880 мкг 2 раза в день)
HFA: 50, 100 или 200 мкг/впрыскивание
< 12 лет: не установлено
≥ 12 лет: так же, как для взрослых
Если ранее применялись только бронхолитики, начальная доза 220 мкг (доставляются 200 мкг) 1 раз в день или 2 раза в день (максимум 440 мкг в день)
Если ранее применялись ингаляционные кортикостероиды, начальная доза 110-220 мкг (доставляется 100 или 200 мкг) 2 раза в день,(максимум 800 мкг в день)
Если ранее применялись пероральные кортикостероиды, начальная доза 440 мкг (доставляется 400 мкг) 2 раза в день (максимум 800 мкг в день)
Системные кортикостероиды (пероральные)
Метилпреднизолон
Таблетки: 2, 4, 8, 16 или 32 мг
0-11 лет: Пульс-терапия: 1-2 мг/кг 1 раз в день (максимум 60 мг) в течение 3-10 дней
≥ 12 лет: так же, как для взрослых
7,5 – 60 мг 1 раз в день утром или через день утром
Пульс-терапия: 40-60 мг 1 раз в день (или 20-30 мг 2 раза в день) в течение 3-10 дней
Поддерживающие дозы — одна доза 1 раз в день (утром, каждый день) или через день.
Согласно данным некоторых исследований, прием препарата в 3 часа дня увеличивает его эффективность, но не сопровождается угнетением функции надпочечников.
Пульс-терапия эффективна для контроля заболевания, если лечение только начинается или наблюдается постепенное ухудшение.
Пульс-терапия продолжается до того момента, пока ПСВ не достигает 80% от лучшего значения для пациента или пока не купируются симптомы (примерно 3-10 дней).
Преднизолон
Таблетки: 5 мг
Раствор: 5 мг/5 мл или 15 мг/5 мл
Преднизон
Таблетки: 1; 2,5; 5; 10; 20 или 50 мг
Раствор: 5 мг/мл или 5 мг/5 мл
Комбинированные препараты
Ипратропия бромид и альбутерол
АРИ: 20 мкг/вдох ипратропия бромида и 100 мкг/вдох альбутерола
Не установлено
1 впрыскивание 4 раза в день (максимум 6 впрыскиваний/день)
Ипратропия бромид пролонгирует бронхолитический эффект альбутерола.
Раствор для небулайзера: 0,5 мг ипратропия бромида и 2,5 мг альбутерола во флаконе 3 мл
Флакон 3 мл 4 раза в день для амбулаторного резервного лечения (максимум 6 доз/24 часа)
—
Флутиказон и сальметерол
Порошковый ингалятор: 100, 250 или 500 мкг флутиказона и 50 мкг сальметерола
< 4 лет: не установлено
4-11 лет: 1 впрыскивание (100/50) 2 раза в день
≥ 12 лет: так же, как для взрослых
1 впрыскивание 2 раза в день
Доза 250/50 используется при отсутствии контроля астмы на низких и средних дозах ингаляционных кортикостероидов.
Доза 500/50 используется при отсутствии контроля астмы на средних и высоких доза ингаляционных кортикостероидов.
ГФА: 45, 115 или 230 мкг флутиказона и 21 мкг сальметерола
< 12 лет: не установлено
≥ 12 лет: так же, как для взрослых
2 впрыскивания 2 раза в день
—
Будесонид и формотерол
ГФА: 80 или 160 мкг будесонида и 4,5 мкг формотерола
< 12 years: 1-2 puffs budesonide 80 mcg/formoterol 4.5 mcg twice daily (not to exceed 2 puffs twice daily as maintenance dose)]
≥ 12 лет: 2 впрыскивания два раза в день и по мере необходимости
Максимальная суточная доза базисной терапии и терапии по требованию составляет 8 ингаляционных доз (36 мкг)
2 ингаляционных дозы 2 раза в день и по требованию Максимальная суточная доза базисной терапии и терапии по требованию составляет 12 ингаляционных доз (54 мкг).
Доза 80/4,5 используется при отсутствии контроля астмы на низких и средних доза ингаляционнных кортикостероидов.
Доза 160/4,5 используется при отсутствии контроля астмы на средних и высоких доза ингаляционных кортикостероидов.
Мометазон и формотерол
HFA: 100 мкг или 200 мкг мометазона и 5 мкг формотерола; 50 мкг мометазона и 5 мкг формотерола для детей < 5 лет
< 5 лет: 2 ингаляционных дозы два раза в день по требованию Максимальная суточная доза базисной те и не превышать эту дозу при базисной терапии
≥ 5 лет: 2 ингаляционных дозы два раза в день по требованию
Максимальная суточная доза базисной терапии и терапии по требованию составляет 8 ингаляционных доз (36 мкг)
2 впрыскивания 2 раза в день и по мере необходимости
Максимальная суточная доза базисной терапии и терапии по требованию составляет 12 ингаляционных доз (54 мкг)
Доза 100/5 рекомендуется при отсутствии контроля астмы на низких и средних дозах ингаляционных кортикостероидов.
Доза 200/5 рекомендуется при отсутствии контроля астмы на высоких доза ингаляционных кортикостероидов.
Флутиказон и вилантерол
Порошковый ингалятор: 100 или 200 мкг флутиказона и 25 мкг вилантерола
Не установлено
1 впрыскивание 1 раз/день
Рекомендуемая начальная доза на основании степени тяжести астмы.
Стабилизаторы тучных клеток
Кромолин (в РФ не зарегистрирован)
Раствор для небулайзера: 20 мг/ампула
< 2 лет: не становлено
≥ 2 лет: так же, как для взрослых
1 ампула 3-4 раза в день
Кромолин следует использовать перед физической нагрузкой или воздействием аллергена.
Одна доза обеспечивает эффективную профилактику в течение 1-2 часов.
Модификаторы лейкотриенов
Монтелукаст
Таблетки, жевательные таблетки, гранулы: 4, 5 или 10 мг
12 месяцев — 5 лет: 4 мг перорально 1 раз в день (вечером);
6-14 лет: 5 мг перорально 1 раз в день (вечером);
≥ 15 лет: так же, как для взрослых
10 мг перорально 1 раз в день (вечером)
Бронхиальная астма физического напряжения: 10 мг перорального за 2 часа до физической нагрузки
Монтелукаст — это антагонист лейкотриеновых рецепторов, конкурентный ингибитор лейкотриенов D4 и E4.
Зафирлукаст
Таблетки: 10 или 20 мг
< 5 лет: не установлено
5–11 лет: 10 мг перорально 2 раза в день
≥ 12 лет: так же, как для взрослых
20 мг перорально вечером
Зафирлукаст — это антагонист лейкотриеновых рецепторов, конкурентный ингибитор лейкотриенов D4 и E4.
Препарат принимают за 1 час до или через 2 часа после еды.
Зилеутон
Таблетки немедленного высвобождения: 600 мг
< 12 лет: не установлено
≥ 12 лет: так же, как для взрослых
600 мг перорально 4 раза в день
Зилеутон — это ингибитор 5-липоксигеназы.
Способ дозирования ограничивает приверженность к терапии.
Зилеутон может вызвать повышение печеночных ферментов и ингибировать метаболизм препаратов, которые расщепляются с помощью CYP3A4, в т.ч. теофиллина.
С пролонгированным высвобождением активного вещества: 1200 мг
< 12 лет: не установлено
≥ 12 лет: так же, как для взрослых
1200 мг перорально 2 раза в день в течение 1 часа после завтрака и ужина
Метилксантины
Теофиллин
Капсулы пролонгированного высвобождения: 100, 200, 300 или 400 мг
Эликсир: 80 мг/15 мл
Таблетки пролонгированного высвобождения: 100, 200, 400, 450 или 600 мг
Начальная доза 10 мг/кг/деньдо 600 мг/день с последующей титрацией дозы до достижения равновесной концентрации в сыворотке крови 5-15 мкг/мл
Начальная доза 10 мг/кг/деньдо 600 мг/день с последующей титрацией дозы до достижения равновесной концентрации в сыворотке крови 5-15 мкг/мл
Большая вариабельность метаболического клиренса, вероятсность лекарственных взаимодействий и нежелательных явлений требует постоянного мониторинга концентрации в сыворотке крови.
В последнее время препарат применяется редко.
Целевой безопасный уровень < 10 мкг/мл.
Иммуномодуляторы
Бенрализумаб
Подкожная инъекция: 30 мг/мл
< 12 лет: не установлено
≥ 12 лет: так же, как для взрослых
30 мг подкожно каждые 4 недели для 3 доз, затем каждые 8 недель после этого
Бенрализумаб используется в качестве дополнительного лечения пациентов с эозинофильным фенотипом астмы.
Дупилумаб
Подкожная инъекция: 300 мг/2 мл или 200 мг/1,14 мл
< 6 лет: не установлено
6-11 лет: обратитесь к специалисту для коррекции дозы
≥ 12 лет: так же, как для взрослых
400 мг подкожно один раз, затем 200 мг каждые 2 недели или 600 мг подкожно один раз, затем 300 мг каждые 2 недели
Начальная доза должна быть дана двумя инъекциями.
Дупилумаб используется в качестве дополнительного лечения пациентов с эозинофильным фенотипом астмы.
Меполизумаб
Подкожная инъекция: 100 мг
< 6 лет: не установлено
6-12 лет: обратитесь к специалисту для коррекции дозы
≥ 12 лет: так же, как для взрослых
100 мг подкожно один раз каждые 4 недели
—
Омализумаб
Подкожные инъекции: 150 мг/1,2 мл
< 12 лет: 75–375 мг подкожно через каждые 2–4 недели, в зависимости от массы тела и уровня IgE в сыворотке перед началом лечения
≥ 12 лет: так же, как для взрослых
150 – 375 мг подкожно через каждые 2–4 недели, в зависимости от массы тела и уровня IgE сыворотки перед началом терапии
Максимальная доза на одну область инъекции — 150 мг.
Реслизумаб
Внутривенно: 100 мг/10 мл
Не установлено
3 мг/кг внутривенно 1 раз каждые 4 недели
—
*Potency expressed as albuterol, equivalent to 3 mg albuterol sulfate.
PRESCRIBING INFORMATION
FOR INHALATION USE ONLY – NOT FOR INJECTION.
DESCRIPTION
Albuterol sulfate inhalation solution is a relatively selective beta2-adrenergic bronchodilator (see CLINICAL PHARMACOLOGY section below). Albuterol sulfate, the racemic form of albuterol, has the chemical name α1-[(tert-Butylamino)methyl]-4-hydroxy-m-xylene-α,α′-diol sulfate (2:1) (salt) and the following structural formula:
Albuterol sulfate has a molecular weight of 576.7, and the molecular formula is (C13H21NO3)2 •H2SO4. Albuterol sulfate is a white or practically white powder, freely soluble in water and slightly soluble in alcohol. The World Health Organization’s recommended name for albuterol base is salbutamol. Albuterol sulfate inhalation solution 0.083% requires no dilution before administration.
Each mL of albuterol sulfate inhalation solution (0.083%) contains 0.83 mg of albuterol (as 1 mg of albuterol sulfate) in an isotonic, sterile, aqueous solution containing sodium chloride; sulfuric acid is used to adjust the pH to between 3 and 5. Albuterol sulfate inhalation solution (0.083%) contains no sulfiting agents. Albuterol sulfate inhalation solution is a clear, colorless solution.
CLINICAL PHARMACOLOGY
The prime action of beta-adrenergic drugs is to stimulate adenyl cyclase, the enzyme which catalyzes the formation of cyclic-3′,5′-adenosine monophosphate (cyclic AMP) from adenosine triphosphate (ATP). The cyclic AMP thus formed mediates the cellular responses. In vitro studies and in vivo pharmacologic studies have demonstrated that albuterol has a preferential effect on beta2-adrenergic receptors compared with isoproterenol. While it is recognized that beta2-adrenergic receptors are the predominant receptors in bronchial smooth muscle, data indicate that 10% to 50% of the beta-receptors in the human heart may be beta2-receptors. The precise function of these receptors, however, is not yet established. Albuterol has been shown in most controlled clinical trials to have more effect on the respiratory tract in the form of bronchial smooth muscle relaxation than isoproterenol at comparable doses while producing fewer cardiovascular effects. Controlled clinical studies and other clinical experience have shown that inhaled albuterol, like other beta-adrenergic agonist drugs, can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or electrocardiographic changes.
Albuterol is longer acting than isoproterenol in most patients by any route of administration because it is not a substrate for the cellular uptake processes for catecholamines nor for catechol-O-methyl transferase.
Studies in asthmatic patients have shown that less than 20% of a single albuterol dose was absorbed following either IPPB (intermittent positive-pressure breathing) or nebulizer administration; the remaining amount was recovered from the nebulizer and apparatus and expired air. Most of the absorbed dose was recovered in the urine 24 hours after drug administration. Following a 3 mg dose of nebulized albuterol, the maximum albuterol plasma level at 0.5 hours was 2.1 ng/mL (range, 1.4 to 3.2 ng/mL).
There was a significant dose-related response in FEV1 (forced expiratory volume in one second) and peak flow rate. It has been demonstrated that following oral administration of 4 mg of albuterol, the elimination half-life was five to six hours.
Animal studies show that albuterol does not pass the blood-brain barrier. Recent studies in laboratory animals (minipigs, rodents, and dogs) recorded the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta-agonists and methylxanthines were administered concurrently. The significance of these findings when applied to humans is currently unknown.
In controlled clinical trials, most patients exhibited an onset of improvement in pulmonary function within 5 minutes as determined by FEV1. FEV1 measurements also showed that the maximum average improvement in pulmonary function usually occurred at approximately 1 hour following inhalation of 2.5 mg of albuterol by compressor-nebulizer and remained close to peak for 2 hours. Clinically significant improvement in pulmonary function (defined as maintenance of a 15% or more increase in FEV1 over baseline values) continued for 3 to 4 hours in most patients and in some patients continued up to 6 hours.
In repetitive dose studies, continued effectiveness was demonstrated throughout the three-month period of treatment in some patients.
Published reports of trials in asthmatic children aged 3 years or older have demonstrated significant improvement in either FEV1 or PEFR within 2 to 20 minutes following a single dose of albuterol inhalation solution. An increase of 15% or more in baseline FEV1 has been observed in children aged 5 to 11 years up to 6 hours after treatment with doses of 0.10 mg/kg or higher of albuterol inhalation solution. Single doses of 3, 4, or 10 mg resulted in improvement in baseline PEFR that was comparable in extent and duration to a 2 mg dose, but doses above 3 mg were associated with heart rate increases of more than 10%.
INDICATIONS AND USAGE
Albuterol sulfate inhalation solution is indicated for the relief of bronchospasm in patients 2 years of age and older with reversible obstructive airway disease and acute attacks of bronchospasm.
CONTRAINDICATIONS
Albuterol sulfate inhalation solution is contraindicated in patients with a history of hypersensitivity to any of its components.
WARNINGS
As with other inhaled beta-adrenergic agonists, albuterol sulfate inhalation solution can produce paradoxical bronchospasm, which can be life threatening. If it occurs, the preparation should be discontinued immediately and alternative therapy instituted.
Fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs and with the home use of nebulizers. It is, therefore, essential that the physician instruct the patient in the need for further evaluation, if his/her asthma becomes worse. In individual patients, any beta2-adrenergic agonist, including albuterol solution for inhalation, may have a clinically significant cardiac effect.
Immediate hypersensitivity reactions may occur after administration of albuterol as demonstrated by rare cases of urticaria, angioedema, rash, bronchospasm, and oropharyngeal edema.
PRECAUTIONS
General
Albuterol, as with all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias and hypertension, in patients with convulsive disorders, hyperthyroidism or diabetes mellitus and in patients who are unusually responsive to sympathomimetic amines.
Large doses of intravenous albuterol have been reported to aggravate pre-existing diabetes mellitus and ketoacidosis. As with other beta-agonists, inhaled and intravenous albuterol may produce a significant hypokalemia in some patients, possibly through intracellular shunting, which has the potential to produce adverse cardiovascular effects. The decrease is usually transient, not requiring supplementation.
Repeated dosing with 0.15 mg/kg of albuterol inhalation solution in children aged 5 to 17 years who were initially normokalemic has been associated with an asymptomatic decline of 20% to 25% in serum potassium levels.
Information for Patients
The action of albuterol sulfate inhalation solution may last up to six hours, and therefore it should not be used more frequently than recommended. Do not increase the dose or frequency of medication without medical consultation. If symptoms get worse, medical consultation should be sought promptly. While taking albuterol sulfate inhalation solution, other anti-asthma medicines should not be used unless prescribed.
Drug compatibility (physical and chemical), efficacy, and safety of albuterol inhalation solution when mixed with other drugs in a nebulizer have not been established.
See illustrated Patient’s Instructions for Use.
Drug Interactions
Other sympathomimetic aerosol bronchodilators or epinephrine should not be used concomitantly with albuterol.
Albuterol should be administered with extreme caution to patients being treated with monoamine oxidase inhibitors or tricyclic antidepressants, since the action of albuterol on the vascular system may be potentiated.
Beta-receptor blocking agents and albuterol inhibit the effect of each other.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Albuterol sulfate caused a significant dose-related increase in the incidence of benign leiomyomas of the mesovarium in a 2-year study in the rat, at oral doses of 2, 10, and 50 mg/kg, corresponding to 10, 50, and 250 times, respectively, the maximum nebulization dose for a 50 kg human. In another study, this effect was blocked by the coadministration of propranolol. The relevance of these findings to humans is not known. An 18-month study in mice and a lifetime study in hamsters revealed no evidence of tumorigenicity. Studies with albuterol revealed no evidence of mutagenesis. Reproduction studies in rats revealed no evidence of impaired fertility.
Pregnancy
Teratogenic Effects
Pregnancy Category C
Albuterol has been shown to be teratogenic in mice when given subcutaneously in doses corresponding to 1.25 times the human nebulization dose (based on a 50 kg human). There are no adequate and well-controlled studies in pregnant women. Albuterol should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. A reproduction study in CD-1 mice with albuterol (0.025, 0.25, and 2.5 mg/kg subcutaneously, corresponding to 0.125, 1.25, and 12.5 times the maximum human nebulization dose, respectively) showed cleft palate formation in 5 of 111 (4.5%) fetuses at 0.25 mg/kg and in 10 of 108 (9.3%) fetuses at 2.5 mg/kg. None were observed at 0.025 mg/kg. Cleft palate also occurred in 22 of 72 (30.5%) fetuses treated with 2.5 mg/kg isoproterenol (positive control). A reproduction study in Stride Dutch rabbits revealed cranioschisis in 7 of 19 (37%) fetuses at 50 mg/kg, corresponding to 250 times the maximum nebulization dose for a 50-kg human.
During worldwide marketing experience, various congenital anomalies, including cleft palate and limb defects, have been rarely reported in the offspring of patients being treated with albuterol. Some of the mothers were taking multiple medications during their pregnancies. No consistent pattern of defects can be discerned, and a relationship between albuterol use and congenital anomalies has not been established.
Labor and Delivery
Oral albuterol has been shown to delay preterm labor in some reports. There are presently no well-controlled studies that demonstrate that it will stop preterm labor or prevent labor at term. Therefore, cautious use of albuterol sulfate inhalation solution is required in pregnant patients when given for relief of bronchospasm so as to avoid interference with uterine contractibility.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because of the potential for tumorigenicity shown for albuterol in some animal studies, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
The safety and effectiveness of albuterol sulfate inhalation solution have been established in children 2 years of age or older. Use of albuterol sulfate inhalation solution in these age groups is supported by evidence from adequate and well-controlled studies of albuterol sulfate inhalation solution in adults; the likelihood that the disease course, pathophysiology, and the drug’s effect in pediatric and adult patients are substantially similar; and published reports of trials in pediatric patients 3 years of age or older. The recommended dose for the pediatric population is based upon three published dose comparison studies of efficacy and safety in children 5 to 17 years, and on the safety profile in both adults and pediatric patients at doses equal to or higher than the recommended doses. The safety and effectiveness of albuterol sulfate inhalation solution in children below 2 years of age have not been established.
ADVERSE REACTIONS
The results of clinical trials with albuterol sulfate inhalation solution in 135 patients showed the following side effects which were considered probably or possibly drug related:
Central Nervous System: tremors (20%), dizziness (7%), nervousness (4%), headache (3%), insomnia (1%).
Gastrointestinal: nausea (4%), dyspepsia (1%).
Ear, Nose and Throat: pharyngitis (<1%), nasal congestion (1%).
Cardiovascular: tachycardia (1%), hypertension (1%).
Respiratory: bronchospasm (8%), cough (4%), bronchitis (4%), wheezing (1%).
No clinically relevant laboratory abnormalities related to albuterol sulfate inhalation solution administration were determined in these studies.
In comparing the adverse reactions reported for patients treated with albuterol sulfate inhalation solution with those of patients treated with isoproterenol during clinical trials of three months, the following moderate to severe reactions, as judged by the investigators, were reported. This table does not include mild reactions.
| Reaction | Albuterol N=65 |
Isoproterenol N=65 |
| Central Nervous System | ||
| Tremors | 10.7% | 13.8% |
| Headache | 3.1% | 1.5% |
| Insomnia | 3.1% | 1.5% |
| Cardiovascular | ||
| Hypertension | 3.1% | 3.1% |
| Arrhythmias | 0% | 3% |
| *Palpitation | 0% | 22% |
| Respiratory | ||
| +Bronchospasm | 15.4% | 18% |
| Cough | 3.1% | 5% |
| Bronchitis | 1.5% | 5% |
| Wheeze | 1.5% | 1.5% |
| Sputum Increase | 1.5% | 1.5% |
| Dyspnea | 1.5% | 1.5% |
| Gastrointestinal | ||
| Nausea | 3.1% | 0% |
| Dyspepsia | 1.5% | 0% |
| Systemic | ||
| Malaise | 1.5% | 0% |
*The finding of no arrhythmias and no palpitations after albuterol administration in the clinical study should not be interpreted as indicating that these adverse effects cannot occur after the administration of inhaled albuterol.
+In most cases of bronchospasm, this term was generally used to describe exacerbations in the underlying pulmonary disease. Cases of urticaria, angioedema, rash, bronchospasm, hoarseness, oropharyngeal edema, arrhythmias (including atrial fibrillations, supraventricular tachycardia, extrasystoles) have been reported after the use of albuterol sulfate inhalation solution.
OVERDOSAGE
Manifestations of overdosage may include seizures, anginal pain, hypertension, hypokalemia, tachycardia with rates up to 200 beats/min, and exaggeration of the pharmacological effects listed in ADVERSE REACTIONS. In isolated cases in children 2 to 12 years of age, tachycardia with rates > 200 beats/min has been observed.
The oral LD50 in rats and mice was greater than 2,000 mg/kg. The inhalation LD50 could not be determined.
There is insufficient evidence to determine if dialysis is beneficial for overdosage of albuterol inhalation solution.
DOSAGE AND ADMINISTRATION
Adults and Children 2 to 12 Years of Age: The usual dosage for adults and for children weighing at least 15 kg is 2.5 mg of albuterol (one vial) administered three to four times daily by nebulization. Children weighing < 15 kg who require < 2.5 mg/dose (i.e., less than a full vial) should use albuterol inhalation solution, 0.5% instead of albuterol inhalation solution, 0.083%. More frequent administration or higher doses are not recommended. To administer 2.5 mg of albuterol, administer the entire contents of one sterile unit-dose vial (3 mL of 0.083% inhalation solution) by nebulization. The flow rate is regulated to suit the particular nebulizer so that albuterol inhalation solution will be delivered over approximately 5 to 15 minutes.
The use of albuterol sulfate inhalation solution can be continued as medically indicated to control recurring bouts of bronchospasm. During this time most patients gain optimum benefit from regular use of the inhalation solution.
If a previously effective dosage regimen fails to provide the usual relief, medical advice should be sought immediately, as this is often a sign of seriously worsening asthma that would require reassessment of therapy.
HOW SUPPLIED
Unit-dose plastic vial containing sterile Albuterol Sulfate Inhalation Solution 0.083%, 2.5 mg* / 3 ml (*Potency expressed as albuterol, equivalent to 3 mg albuterol sulfate). Equivalent to 0.5 mL of albuterol sulfate inhalation solution, 0.5% diluted to 3 mL with normal saline. Supplied in cartons as listed below.
NDC 21695-332-25 25 vials per carton
NDC 21695-332-30 30 vials per carton
Storage and Handling
PROTECT FROM LIGHT. Store vials in pouch until time of use. Store between 2° and 25° C (36° and 77° F).
Rx Only
Manufactured By and For:
Nephron Pharmaceuticals Corporation
Orlando, FL 32811
For Customer Service,
Call 1-800-443-4313
Repackaged by:
Rebel Distributors Corp.
Thousand Oaks, CA 91320
Patient Package Insert
Albuterol Sulfate Inhalation Solution, 0.083%*
2.5 mg*/3 mL
*Potency expressed as albuterol, equivalent to 3 mg albuterol sulfate.
Note: This is a unit-dose vial. No dilution is required. Read complete instructions carefully before using.
INSTRUCTIONS FOR USE
- Remove vial from the foil pouch.
- Twist the cap completely off the vial and squeeze the contents into the nebulizer reservoir (Figure 1).
-
Figure 1
- Connect the nebulizer reservoir to the mouthpiece or face mask (Figure 2).
-
Figure 2
- Connect the nebulizer to the compressor.
- Sit in a comfortable, upright position; place the mouthpiece in your mouth (Figure 3)(or put on the face mask); and turn on the compressor.
-
Figure 3
- Breathe as calmly, deeply and evenly as possible until no more mist is formed in the nebulizer chamber (about 5 to 15 minutes). At this point, the treatment is finished.
- Clean the nebulizer (see manufacturer’s instructions).
Note: Use only as directed by your physician. More frequent administration or higher doses are not recommended.
Store Albuterol Sulfate Inhalation Solution, 0.083%* between 2° and 25° C (36° and 77° F). Store vials in pouch until time of use.
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Principal Display Panel –Label (25 Count)
NDC 21695-332-25
Albuterol Sulfate Inhalation Solution, 0.083%*
2.5 mg*/3 mL
*Potency expressed as albuterol, equivalent to 3 mg albuterol sulfate.
FOR ORAL INHALATION ONLY
Equivalent to 0.5 mL Albuterol Sulfate 0.5%* diluted to 3 mL with normal saline.
Each mL contains 1 mg albuterol sulfate, equivalent to 0.83 mg albuterol in an aqueous solution containing sodium chloride, and sulfuric acid to adjust pH between 3 and 5. Contains no preservatives.
Attention Pharmacist: Detach “Patient’s Instructions For Use” from package insert and dispense with solution.
Please consult your physician before use. Do not exceed recommended dosage.
Usual Dosage: See package insert.
Protect from light. Store between 2° and 25° C (36° and 77° F).
Discard if solution becomes discolored. (Note: Albuterol Sulfate Inhalation Solution is a clear, colorless to light yellow solution.)
Rx Only.
Sterile
3 mL Unit-Dose Vials
Manufactured By:
Nephron Pharmaceuticals Corporation
Orlando, FL 32811
Repackaged by:
Rebel Distributors Corp.
Thousand Oaks, CA 91320
Principal Display Panel –Carton (25 count)
Rebel Distributors Corp.