Состав:
Применение:
Применяется при лечении:
Страница осмотрена фармацевтом Федорченко Ольгой Валерьевной Последнее обновление 2022-04-05
Внимание!
Информация на странице предназначена только для медицинских работников!
Информация собрана в открытых источниках и может содержать значимые ошибки!
Будьте внимательны и перепроверяйте всю информацию с этой страницы!
Топ 20 лекарств с такими-же компонентами:
Топ 20 лекарств с таким-же применением:
Предоставленная в разделе Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
Vilerm
Состав
Предоставленная в разделе Состав Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Состав
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
Aciclovir
Терапевтические показания
Предоставленная в разделе Терапевтические показания Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Терапевтические показания
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
Крем для наружного применения; Драже; Порошок для приготовления раствора для инфузий
Мазь; Таблетки
Острые герпетические заболевания кожи и слизистых оболочек (вызванные вирусом простого герпеса), включая первичный и рецидивирующий герпес половых органов, неонатальный герпес; герпетические инфекции любой локализации у больных с иммунодефицитом (лечение и профилактика); опоясывающий лишай, ветряная оспа.
Для системного применения:
— лечение инфекций кожи и слизистых оболочек, вызванных вирусами Herpes simplex типа I и II (первичные и вторичные, включая генитальный герпес);
— профилактика обострений рецидивирующих инфекций, вызванных вирусами Herpes simplex типа I и II, у пациентов с нормальным иммунным статусом;
— профилактика первичных и рецидивирующих инфекций, вызванных вирусами Herpes simplex типа I и II, у больных с иммунодефицитом;
— в составе комплексной терапии пациентов с выраженным иммунодефицитом: при ВИЧ-инфекции (стадия СПИДа, ранние клинические проявления и развернутая клиническая картина) и у пациентов, перенесших трансплантацию костного мозга;
— лечение первичных и рецидивирующих инфекций, вызванных вирусом Varicella zoster (ветряная оспа, опоясывающий лишай).
Для наружного применения: простой герпес кожи, генитальный герпес (простой и рецидивирующий), лабиальный герпес; опоясывающий лишай и ветряная оспа.
Способ применения и дозы
Предоставленная в разделе Способ применения и дозы Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Способ применения и дозы
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
Крем для наружного применения; Драже; Порошок для приготовления раствора для инфузий
Мазь; Таблетки
Таблетки, покрытые пленочной оболочкой
Внутрь: по 200 мг 4–5 раз в сутки при простом герпесе, герпесе половых органов; по 400 мг 5 раз в сутки для профилактики герпеса у пациентов, которым проводится активная иммуносупрессивная терапия (при трансплантации органов); по 400–800 мг 5 раз в сутки при опоясывающем лишае.
В/в: по 15–30 мг/кг/сут при первичном генитальном герпесе, неонатальном герпесе, опоясывающем лишае, ветряной оспе у больных с врожденным нарушением иммунного ответа или вторичными иммунными дефектами; детям от 3 мес до 12 лет — 250–500 мг/м2 /сут.
Накожно (крем): 5 раз в сутки (каждые 4 ч) наносят на пораженные участки кожи для снижения боли и отека при повторных высыпаниях герпеса на гениталиях и лице.
Внутрь, во время или сразу после приема пищи, запивая достаточным количеством воды. При лечении инфекций кожи и слизистых оболочек, вызванных Herpes simplex I или II, взрослым и детям старше 2 лет — по 200 мг 5 раз в сутки в течение 5 дней с 4 ч интервалами в течении дня и с 8 ч интервалом на ночь (при необходимости курс может быть продлен). В составе комплексной терапии при выраженном иммунодефиците (в т.ч. при развернутой клинической картине ВИЧ-инфекции, включая ранние клинические проявления ВИЧ-инфекции и СПИДа), после трансплантации костного мозга — по 400 мг 5 раз в сутки.
Для профилактики рецидивов инфекций, вызванных Herpes simplex I или II, пациентам с нормальным иммунным статусом и при рецидиве заболевания — по 200 мг 4 раза в сутки каждые 6 ч (максимальная доза — до 400 мг 5 раз в сутки в зависимости от тяжести инфекции).
При лечении инфекций, вызванных Varicella zoster, взрослым и детям массой более 40 кг — по 800 мг 5 раз в сутки каждые 4 ч днем и с 8 ч интервалом на ночь. Продолжительность лечения — 7–10 дней. Детям старше 2 лет — по 20 мг/кг 4 раза в сутки в течение 5 дней.
При лечении инфекций, вызванных Herpes zoster, взрослым — по 800 мг 4 раза в сутки каждые 6 ч днем в течение 5 дней.
При нарушении функции почек при лечении и профилактике инфекций, вызванных Herpes simplex, у больных с Cl креатинина менее 10 мл/мин дозу препарата снижают до 200 мг 2 раза в сутки с 12 ч интервалом.
При лечении и профилактике инфекций, вызванных Varicella zoster, у больных с Cl креатинина менее 10 мл/мин дозу препарата снижают до 800 мг 2 раза в сутки с 12 ч интервалом, при Cl креатинина до 25 мл/мин — по 800 мг 3 раза в сутки с 8 ч интервалом.
Наружно, мазь наносят на пораженную поверхность 5 раз в сутки (через 4 ч). Продолжительность лечения — 5–10 дней.
Наружно. Препарат наносят тонким слоем на пораженные и граничащие с ними участки кожи 5 раз в день (каждые 4 ч, за исключением ночного времени). Крем наносят ватным тампоном либо чистыми руками, чтобы избежать дополнительного инфицирования пораженных участков. Важно начать лечение как можно раньше, желательно при появлении первых признаков и симптомов (в продромальном периоде или при покраснении). Лечение также может начаться на более поздних стадиях (папулы или пузыри).
Терапию следует продолжать до того времени, пока на пузырьках не образуется корка, либо пока они полностью не заживут. Длительность лечения — не менее 5 дней, лечение может быть продолжено до 10 дней. В случае сохранения симптомов заболевания более 10 дней следует обратиться к врачу.
Противопоказания
Предоставленная в разделе Противопоказания Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Противопоказания
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
Крем для наружного применения; Драже; Порошок для приготовления раствора для инфузий
Мазь; Таблетки
Гиперчувствительность.
Гиперчувствительность, в т.ч. к ацикловиру или компонентам препарата, кормление грудью; с осторожностью — дегидратация, почечная недостаточность, неврологические нарушения, в т.ч. в анамнезе.
Побочные эффекты
Предоставленная в разделе Побочные эффекты Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Побочные эффекты
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
Крем для наружного применения; Драже; Порошок для приготовления раствора для инфузий
Мазь; Таблетки
Диспептические явления, головная боль, головокружение, аллергические реакции.
При приеме внутрь.
Со стороны нервной системы и органов чувств: редко — головная боль, слабость; в отдельных случаях — тремор, головокружение, повышенная утомляемость, сонливость, галлюцинации.
Со стороны органов ЖКТ: в единичных случаях — боль в животе, тошнота, рвота, диарея.
Аллергические реакции: кожная сыпь.
Прочие: транзиторное повышение активности ферментов печени; редко — повышение уровня мочевины и креатинина, гипербилирубинемия, лейкопения, эритропения, алопеция, лихорадка.
При наружном применении: возможно покраснение, зуд, шелушение, жжение или покалывание в месте аппликации; в редких случаях — аллергический дерматит.
Передозировка
Предоставленная в разделе Передозировка Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Передозировка
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
Симптомы: при в/в введении — повышение уровня сывороточного креатинина, азота мочевины крови, почечная недостаточность, неврологические симптомы (спутанность сознания, галлюцинации, возбуждение, судороги и кома).
Лечение: гемодиализ значительно усиливает выведение ацикловира из крови и может быть оптимальным методом лечения при его передозировке.
Нет данных о передозировке при использовании в виде глазной мази.
Фармакодинамика
Предоставленная в разделе Фармакодинамика Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Фармакодинамика
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
Обладает высокой специфичностью в отношении вируса простого герпеса (Herpes simplex) типа I и II, вируса, вызывающего ветряную оспу и опоясывающий лишай (Varicella zoster), вируса Эпштейна — Барр и цитомегаловируса.
Фармакокинетика
Предоставленная в разделе Фармакокинетика Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Фармакокинетика
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
При приеме внутрь биодоступность составляет 15–30%, Cmax достигается через 1,5–2 ч. Концентрация после назначения внутрь 200 мг 5 раз в сутки составляет 0,7 мкг/мл. Связывание с белками плазмы составляет 9–33% и не зависит от концентрации ацикловира в плазме. Хорошо проникает во все органы и ткани, включая головной мозг и кожу. Концентрация в спинномозговой жидкости составляет 50% от концентрации в плазме. Проходит через плацентарный барьер, накапливается в грудном молоке. Метаболизируется в печени с образованием фармакологически неактивного производного — 9-карбоксиметоксиметилгуанина. T1/2 у взрослых с нормальной функцией почек — 2–3 ч, при тяжелой почечной недостаточности — 20 ч, у больных при гемодиализе — 5,7 ч (при этом концентрация ацикловира в плазме уменьшается до 60% от исходного значения). Выводится почками (84% — в неизмененном виде, 14% — в виде метаболита), менее 2% — через кишечник. Почечный клиренс составляет 75–80% от общего клиренса.
Фармокологическая группа
Предоставленная в разделе Фармокологическая группа Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Фармокологическая группа
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
- Противовирусное средство [Противовирусные (за исключением ВИЧ) средства]
Взаимодействие
Предоставленная в разделе Взаимодействие Vilermинформация составлена на основе данных о другом лекарстве с точно таким же составом как лекарство Vilerm. Будьте
внимательны и обязательно уточняйте информацию по разделу Взаимодействие
в инструкции к лекарству Vilerm непосредственно из упаковки или у фармацевта в аптеке.
more…
При приеме внутрь: пробенецид увеличивает средний T1/2 и снижает клиренс ацикловира, нефротоксичные препараты повышают риск нарушения функции почек. При наружном применении: взаимодействия с др. ЛС не выявлено.
Vilerm цена
У нас нет точных данных по стоимости лекарства.
Однако мы предоставим данные по каждому действующему веществу
Средняя стоимость Aciclovir 800 mg за единицу в онлайн аптеках от 0.69$ до 1.08$, за упаковку от 38$ до 73$.
Средняя стоимость Aciclovir 200 mg за единицу в онлайн аптеках от 0.53$ до 1.2$, за упаковку от 30$ до 40$.
Средняя стоимость Aciclovir 400 mg за единицу в онлайн аптеках от 0.62$ до 0.62$, за упаковку от 35$ до 35$.
Источники:
- https://www.drugs.com/search.php?searchterm=vilerm
- https://pubmed.ncbi.nlm.nih.gov/?term=vilerm
Доступно в странах
Найти в стране:
А
Б
В
Г
Д
Е
З
И
Й
К
Л
М
Н
О
П
Р
С
Т
У
Ф
Х
Ч
Ш
Э
Ю
Я
- Описание
- Отзывы (0)
- Доставка и оплата
Показания к применению таблеток
- простой герпес (активная стадия, высыпания, прогрессирование заболевания)
- генитальный герпес
- опоясывающий лишай
- ветряная оспа
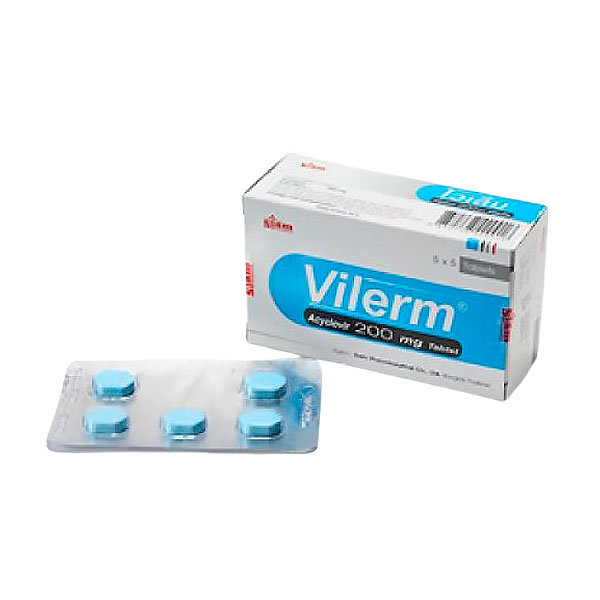
Тайский ацикловир Vilerm
Siam Pharmaceutical Vilerm Acyclovir 200mg.
Таблетки Vilerm от тайского производителя Siam Pharmaceutical – это аптечный противовирусный препарат на основе ацикловира, проверенное средство против герпеса.
Активный компонент таблеток Vilerm – 0,2 гр ацикловир, противовирусное антигерпетическое средство.
Таблетки от герпеса, тайский ацикловир Vilerm показали свою эффективность:
- При простом герпесе кожи и слизистых оболочек (первичный и рецидивирующий)
- При генитальном герпесе (первичный и рецидивирующий)
- При опоясывающем лишае (опоясывающий герпес)
- При ветряной оспе в первые 24 часа после появления типичной сыпи
Способ применения
Способ применения и дозировка указаны в аннотации внутри упаковки и зависят от заболевания и/или интенсивности его течения
Противопоказания
Индивидуальная непереносимость
Объем: 5 блистеров по 5 таблеток (25 таблеток)
Производство: Siam Pharmaceutical, Vilerm, Таиланд
Пищевая добавка не является лекарственным средством, требуется консультация специалиста.
ДОСТАВКА
Все товары отправляются только из Тайланда
Выдача посылки в ближайшем почтовом отделении
Доставку товаров осуществляют почтовые службы Таиланда и Почта России
Срок доставки
Посылки отправляются авиапочтой, срок доставки зависит от региона:
- Центральный, Северо-Западный, Южный, Приволжский, Северо-Кавказский (Москва, Санкт-Петербург, Ростов-на-Дону, Нижний Новгород, Тверь, Сратов, Пятигорск, Анапа) — 7-14 дней
- Уральский, Сибирский (Екатеринбург, Пермь, Красноярск, Новосибирск, Омск, ) — 10-21 день
- Дальневосточный (Владивосток, Южно-Сахалинск, Улан-Удэ) — 14-21 день
ОПЛАТА
Оплата любым удобным способом
Обращаем Ваше внимание, что мы не отправляем заказы наложенным платежом, оплата при получении не возможна!
Отправка происходит только после 100% оплаты заказа!
Оплата через мобильное приложение банка
Вы можете оплатить заказ по номеру карты (Сбер) через мобильное приложение или на сайте банка
Оплата онлайн
Оплата онлайн осуществляется через платежный шлюз Юкасса по защищенному HTTPS протоколу.
Vilerm Aciclovir
- Siam Pharmaceutical Vilerm Acyclovir 200 mg
- Таблетки Vilerm от тайского производителя Siam Pharmaceutical – это аптечный противовирусный препарат на основе ацикловира, проверенное средство против герпеса
- Активный компонент таблеток Vilerm – 0,2 гр ацикловир, противовирусное антигерпетическое средство
Показания к применению таблеток
- простой герпес (активная стадия, высыпания, прогрессирование заболевания)
- генитальный герпес
- опоясывающий лишай
- ветряная оспа
Эффект от применения
Снижает количество активно циркулирующего вируса и убирает герпетические высыпания при:
- герпесе кожи и слизистых оболочек (первичный и рецидивирующий)
- генитальном герпесе (первичный и рецидивирующий)
- опоясывающем лишае (опоясывающий герпес)
- ветряной оспе в первые 24 часа после появления типичной сыпи
Способ применения
Способ применения и дозировка указаны в аннотации внутри упаковки и зависят от заболевания и/или интенсивности его течения
Противопоказания
Индивидуальная непереносимость
Побочные действия
При соблюдении рекомендованной дозы отсутствуют
Особые указания
Хранить в местах, недоступных детям
Избегать передозировки
Способ хранения
Хранить в сухом прохладном месте
Беречь от попадания прямого солнечного света
Состав
в каждой таблетке – 0,2 гр. ацикловир
Срок годности
Указан производителем на упаковке
Объем: 5 блистеров по 5 таблеток (25 таблеток)
Производство: Siam Pharmaceutical, Vilerm, Таиланд
Препарат сертифицирован и продается в аптеках Таиланда без рецепта. Перед применением требуется консультация специалиста.
Components:
Method of action:
Treatment option:
Medically reviewed by Fedorchenko Olga Valeryevna, PharmD. Last updated on 2022-04-05
Attention!
Information on this page is intended only for medical professionals!
Information is collected in open sources and may contain significant errors!
Be careful and double-check all the information on this page!
Top 20 medicines with the same components:
Top 20 medicines with the same treatments:
The information provided in of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Vilerm
Qualitative and quantitative composition
The information provided in Qualitative and quantitative composition of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Qualitative and quantitative composition in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Aciclovir
Therapeutic indications
The information provided in Therapeutic indications of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Therapeutic indications in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
Vilerm Tablets are indicated for the treatment of herpes simplex virus infections of the skin and mucous membranes including initial and recurrent genital herpes (excluding neonatal HSV and severe HSV infections in immunocompromised children).
Vilerm Tablets are indicated for the suppression (prevention of recurrences) of recurrent herpes simplex infections in immunocompetent patients.
Vilerm Tablets are indicated for the prophylaxis of herpes simplex infections in immunocompromised patients.
Vilerm Tablets are indicated for the treatment of varicella (chickenpox) and herpes zoster (shingles) infections.
Vilerm Tablets are indicated for the treatment of herpes simplex virus infections of the skin and mucous membranes including initial and recurrent genital herpes (excluding neonatal HSV and severe HSV infections in immunocompromised children).
Vilerm Tablets are indicated for the suppression (prevention of recurrences) of recurrent herpes simplex infections in immunocompetent patients.
Vilerm Tablets are indicated for the prophylaxis of herpes simplex infections in immunocompromised patients.
Vilerm Tablets are indicated for the treatment of varicella (chickenpox) and herpes zoster (shingles) infections.
Route of administration: Oral.
Dosage (Posology) and method of administration
The information provided in Dosage (Posology) and method of administration of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Dosage (Posology) and method of administration in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
Posology
Dosage in adults
Treatment of herpes simplex infections: 200 mg Vilerm should be taken five times daily at approximately four hourly intervals omitting the night time dose. Treatment should continue for 5 days, but in severe initial infections this may have to be extended.
In severely immunocompromised patients (e.g. after marrow transplant) or in patients with impaired absorption from the gut the dose can be doubled to 400 mg Vilerm, or alternatively, intravenous dosing could be considered.
Dosing should begin as early as possible after the start of an infection; for recurrent episodes this should preferably be during the prodromal period or when lesions first appear.
Suppression of herpes simplex infections in immunocompetent patients: 200 mg Vilerm should be taken four times daily at approximately six-hourly intervals.
Many patients may be conveniently managed on a regimen of 400 mg Vilerm twice daily at approximately twelve-hourly intervals.
Dosage titration down to 200 mg Vilerm taken thrice daily at approximately eight-hourly intervals or even twice daily at approximately twelve-hourly intervals may prove effective.
Some patients may experience break-through infection on total daily doses of 800 mg Vilerm.
Therapy should be interrupted periodically at intervals of six to twelve months, in order to observe possible changes in the natural history of the disease.
Prophylaxis of herpes simplex infections in immunocompromised patients: 200 mg Vilerm should be taken four times daily at approximately six-hourly intervals.
In severely immunocompromised patients (e.g. after marrow transplant) or in patients with impaired absorption from the gut, the dose can be doubled to 400 mg Vilerm, or alternatively, intravenous dosing could be considered.
The duration of prophylactic administration is determined by the duration of the period at risk.
Treatment of varicella and herpes zoster infections: 800 mg Vilerm should be taken five times daily at approximately four-hourly intervals, omitting the night time dose. Treatment should continue for seven days.
In severely immunocompromised patients (e.g. after marrow transplant) or in patients with impaired absorption from the gut, consideration should be given to intravenous dosing.
Dosing should begin as early as possible after the start of an infection: Treatment of herpes zoster yields better results if initiated as soon as possible after the onset of the rash. Treatment of chickenpox in immunocompetent patients should begin within 24 hours after onset of the rash.
Paediatric population
Treatment of herpes simplex infections, and prophylaxis of herpes simplex infections in the immunocompromised: Children aged two years and over should be given adult dosages and children below the age of two years should be given half the adult dose.
For treatment on neonatal herpes virus infections, intravenous Vilerm is recommended.
Treatment of varicella infection
6 years and over: 800 mg Vilerm four times daily
2 — 5 years: 400 mg Vilerm four times daily
Under 2 years: 200 mg Vilerm four times daily
Treatment should continue for five days.
Dosing may be more accurately calculated as 20 mg/kg bodyweight (not to exceed 800 mg) Vilerm four times daily.
No specific data are available on the suppression of herpes simplex infections or the treatment of herpes zoster infections in immunocompetent children.
Dosage in the elderly
The possibility of renal impairment in the elderly must be considered and the dosage should be adjusted accordingly (see Dosage in renal impairment below). Adequate hydration of elderly patients taking high oral doses of Vilerm should be maintained.
Dosage in renal impairment
Caution is advised when administering Vilerm to patients with impaired renal function. Adequate hydration should be maintained.
In the management of herpes simplex infections in patients with impaired renal function, the recommended oral doses will not lead to accumulation of Vilerm above levels that have been established safe by intravenous infusion. However for patients with severe renal impairment (creatinine clearance less than 10 ml/minute) an adjustment of dosage to 200 mg Vilerm twice daily at approximately twelve-hourly intervals is recommended.
In the treatment of herpes zoster infections it is recommended to adjust the dosage to 800 mg Vilerm twice daily at approximately twelve hourly intervals for patients with severe renal impairment (creatinine clearance less than 10 ml/minute), and to 800 mg Vilerm three times daily at intervals of approximately eight hours for patients with moderate renal impairment (creatinine clearance in the range 10 — 25 ml/minute).
Method of administration:
Oral.
Vilerm tablets may be dispersed in a minimum of 50 ml of water or swallowed whole with a little water. Ensure that patients on high doses of Vilerm are adequately hydrated.
Vilerm tablets may be dispersed in a minimum of 50 ml of water or swallowed whole with a little water. Ensure that patients on high doses of aciclovir are adequately hydrated.
Dosage in adults
Treatment of herpes simplex infections: 200 mg Vilerm should be taken five times daily at approximately four hourly intervals omitting the night time dose. Treatment should continue for 5 days, but in severe initial infections this may have to be extended.
In severely immunocompromised patients (e.g. after marrow transplant) or in patients with impaired absorption from the gut the dose can be doubled to 400 mg Vilerm or alternatively intravenous dosing could be considered.
Dosing should begin as early as possible after the start of an infection; for recurrent episodes this should preferably be during the prodromal period or when lesions first appear.
Suppression of herpes simplex infections in immunocompetent patients: 200 mg Vilerm should be taken four times daily at approximately six-hourly intervals.
Many patients may be conveniently managed on a regimen of 400 mg Vilerm twice daily at approximately twelve-hourly intervals.
Dosage titration down to 200 mg Vilerm taken thrice daily at approximately eight-hourly intervals or even twice daily at approximately twelve-hourly intervals may prove effective.
Some patients may experience break-through infection on total daily doses of 800 mg Vilerm.
Therapy should be interrupted periodically at intervals of six to twelve months, in order to observe possible changes in the natural history of the disease.
Prophylaxis of herpes simplex infections in immunocompromised patients: 200 mg Vilerm should be taken four times daily at approximately six-hourly intervals.
In severely immunocompromised patients (e.g. after marrow transplant) or in patients with impaired absorption from the gut, the dose can be doubled to 400 mg Vilerm, or alternatively, intravenous dosing could be considered.
The duration of prophylactic administration is determined by the duration of the period at risk.
Treatment of varicella and herpes zoster infections: 800 mg Vilerm should be taken five times daily at approximately four-hourly intervals, omitting the night time dose. Treatment should continue for seven days.
In severely immunocompromised patients (e.g. after marrow transplant) or in patients with impaired absorption from the gut, consideration should be given to intravenous dosing.
Dosing should begin as early as possible after the start of an infection: Treatment of herpes zoster yields better results if initiated as soon as possible after the onset of the rash. Treatment of chickenpox in immunocompetent patients should begin within 24 hours after onset of the rash.
Dosage in children
Treatment of herpes simplex infections, and prophylaxis of herpes simplex infections in the immunocompromised: Children aged two years and over should be given adult dosages and children below the age of two years should be given half the adult dose.
For treatment on neonatal herpes virus infections, intravenous aciclovir is recommended.
Treatment of varicella infection
|
6 years and over: |
800 mg Vilerm four times daily. |
|
2 — 5 years: |
400mg Vilerm four times daily. |
|
Under 2 years: |
200mg Vilerm four times daily. |
Treatment should continue for five days.
Dosing may be more accurately calculated as 20 mg/kg bodyweight (not to exceed 800 mg) Vilerm four times daily.
No specific data are available on the suppression of herpes simplex infections or the treatment of herpes zoster infections in immunocompetent children.
Dosage in the elderly:
The possibility of renal impairment in the elderly must be considered and the dosage should be adjusted accordingly (see Dosage in renal impairment below).
Adequate hydration of elderly patients taking high oral doses of aciclovir should be maintained.
Dosage in renal impairment:
Caution is advised when administering aciclovir to patients with impaired renal function. Adequate hydration should be maintained.
In the management of herpes simplex infections in patients with impaired renal function, the recommended oral doses will not lead to accumulation of aciclovir above levels that have been established safe by intravenous infusion. However for patients with severe renal impairment (creatinine clearance less than 10 ml/minute) an adjustment of dosage to 200 mg aciclovir twice daily at approximately twelve-hourly intervals is recommended.
In the treatment of herpes zoster infections it is recommended to adjust the dosage to 800 mg aciclovir twice daily at approximately twelve — hourly intervals for patients with severe renal impairment (creatinine clearance less than 10 ml/minute), and to 800 mg aciclovir three times daily at intervals of approximately eight hours for patients with moderate renal impairment (creatinine clearance in the range 10 — 25 ml/minute).
Contraindications
The information provided in Contraindications of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Contraindications in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Special warnings and precautions for use
The information provided in Special warnings and precautions for use of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Special warnings and precautions for use in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
Use in patients with renal impairment and in elderly patients:
Vilerm is eliminated by renal clearance, therefore the dose must be adjusted in patients with renal impairment (see 4.2 Posology and Method of Administration).
Elderly patients are likely to have reduced renal function and therefore the need for dose adjustment must be considered in this group of patients. Both elderly patients and patients with renal impairment are at increased risk of developing neurological side effects and should be closely monitored for evidence of these effects. In the reported cases, these reactions were generally reversible on discontinuation of treatment (see 4.8 Undesirable Effects).
Prolonged or repeated courses of Vilerm in severely immune-compromised individuals may result in the selection of virus strains with reduced sensitivity, which may not respond to continued Vilerm treatment.
Hydration status: Care should be taken to maintain adequate hydration in patients receiving high oral doses of Vilerm.
The risk of renal impairment is increased by use with other nephrotoxic drugs.
The data currently available from clinical studies is not sufficient to conclude that treatment with Vilerm reduces the incidence of chickenpox-associated complications in immunocompetent patients.
Paediatric population:
Oral Vilerm should be used in paediatric population mainly for the treatment of non-severe skin and mucosa HSV infections. For the treatment of neonatal HSV and severe HSV infections in immunocompromised children IV Vilerm should be used.
Use in patients with renal impairment and in elderly patients:
Aciclovir is eliminated by renal clearance, therefore the dose must be adjusted in patients with renal impairment (see 4.2 Posology and Method of Administration). Elderly patients are likely to have reduced renal function and therefore the need for dose adjustment must be considered in this group of patients. Both elderly patients and patients with renal impairment are at increased risk of developing neurological side effects and should be closely monitored for evidence of these effects. In the reported cases, these reactions were generally reversible on discontinuation of treatment (see 4.8 Undesirable Effects). Prolonged or repeated courses of aciclovir in severely immune-compromised individuals may result in the selection of virus strains with reduced sensitivity, which may not respond to continued aciclovir treatment.
Hydration status: Care should be taken to maintain adequate hydration in patients receiving high oral doses of aciclovir.
The risk of renal impairment is increased by use with other nephrotoxic drugs.
The data currently available from clinical studies is not sufficient to conclude that treatment with aciclovir reduces the incidence of chickenpox-associated complications in immunocompetent patients.
Effects on ability to drive and use machines
The information provided in Effects on ability to drive and use machines of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Effects on ability to drive and use machines in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
There have been no studies to investigate the effect of Vilerm on driving performance or the ability to operate machinery. A detrimental effect on such activities cannot be predicted from the pharmacology of the active substance, but the adverse event profile should be borne in mind.
There have been no studies to investigate the effect of aciclovir on driving performance or the ability to operate machinery. A detrimental effect on such activities cannot be predicted from the pharmacology of the active substance, but the adverse event profile should be borne in mind.
Undesirable effects
The information provided in Undesirable effects of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Undesirable effects in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
).
Prolonged or repeated courses of Vilerm in severely immune-compromised individuals may result in the selection of virus strains with reduced sensitivity, which may not respond to continued Vilerm treatment.
Hydration status: Care should be taken to maintain adequate hydration in patients receiving high oral doses of Vilerm.
The risk of renal impairment is increased by use with other nephrotoxic drugs.
The data currently available from clinical studies is not sufficient to conclude that treatment with Vilerm reduces the incidence of chickenpox-associated complications in immunocompetent patients.
Paediatric population:
Oral Vilerm should be used in paediatric population mainly for the treatment of non-severe skin and mucosa HSV infections. For the treatment of neonatal HSV and severe HSV infections in immunocompromised children IV Vilerm should be used.
4.5 Interaction with other medicinal products and other forms of interaction
Vilerm is eliminated primarily unchanged in the urine via active renal tubular secretion. Any drugs administered concurrently that compete with this mechanism may increase Vilerm plasma concentrations.
Probenecid and cimetidine increase the AUC of Vilerm by this mechanism, and reduce Vilerm renal clearance. Similarly increases in plasma AUCs of Vilerm and of the inactive metabolite of mycophenolate mofetil, an immunosuppresant agent used in transplant patients have been shown when the drugs are co administered. However no dosage adjustment is necessary because of the wide therapeutic index of Vilerm.
An experimental study on five male subjects indicates that concomitant therapy with Vilerm increases AUC of totally administered theophylline with approximately 50%. It is recommended to measure plasma concentrations during concomitant therapy with Vilerm.
4.6 Fertility, pregnancy and lactation
Pregnancy
The use of Vilerm should be considered only when the potential benefits outweigh the possibility of unknown risks. A post-marketing Vilerm pregnancy registry has documented pregnancy outcomes in women exposed to any formulation of Vilerm. The registry findings have not shown an increase in the number of birth defects amongst Vilerm exposed subjects compared with the general population, and any birth defects showed no uniqueness or consistent pattern to suggest a common cause. Systemic administration of Vilerm in internationally accepted standard tests did not produce embryotoxic or teratogenic effects in rabbits, rats or mice. In a non-standard test in rats, foetal abnormalities were observed but only following such high subcutaneous doses that maternal toxicity was produced. The clinical relevance of these findings is uncertain.
Caution should however be exercised by balancing the potential benefits of treatment against any possible hazard.
Breastfeeding
Following oral administration of 200 mg Vilerm five times a day, Vilerm has been detected in breast milk at concentrations ranging from 0.6 to 4.1 times the corresponding plasma levels. These levels would potentially expose nursing infants to Vilerm dosages of up to 0.3 mg/kg/day. Caution is therefore advised if Vilerm is to be administered to a nursing woman.
Fertility
There is no information on the effect of Vilerm on human female fertility.
In a study of 20 male patients with normal sperm count, oral Vilerm administered at doses of up to 1g per day for up to six months has been shown to have no clinically significant effect on sperm count, motility or morphology.
4.7 Effects on ability to drive and use machines
There have been no studies to investigate the effect of Vilerm on driving performance or the ability to operate machinery. A detrimental effect on such activities cannot be predicted from the pharmacology of the active substance, but the adverse event profile should be borne in mind.
4.8 Undesirable effects
The frequency categories associated with the adverse events below are estimates. For most events, suitable data for estimating incidence were not available. In addition, adverse events may vary in their incidence depending on the indication.
The following convention has been used for the classification of undesirable effects in terms of frequency: Very common > 1/10, common > 1/100 and < 1/10, uncommon > 1/1000 and < 1/100, rare > 1/10,000 and < 1/1000, very rare < 1/10,000
Blood and the lymphatic system disorders:
Very rare: Anaemia, leukopenia, thrombocytopenia.
Immune system disorders:
Rare: Anaphylaxis.
Psychiatric and nervous system disorders:
Common: Headache, dizziness.
Very rare: Agitation, confusion, tremor, ataxia, dysarthria, hallucinations, psychotic symptoms, convulsions, somnolence, encephalopathy, coma.
The above events are generally reversible and usually reported in patients with renal impairment or with other predisposing factors (see 4.4 Special Warnings and Precautions for Use).
Respiratory, thoracic and mediastinal disorders:
Rare: Dyspnoea.
Gastrointestinal disorders:
Common: Nausea, vomiting, diarrhoea, abdominal pains.
Hepato-biliary disorders:
Rare: Reversible rises in bilirubin and liver related enzymes.
Very rare: Hepatitis, jaundice.
Skin and subcutaneous tissue disorders:
Common: Pruritus, rashes (including photosensitivity).
Uncommon: Urticaria. Accelerated diffuse hair loss. Accelerated diffuse hair loss has been associated with a wide variety of disease processes and medicines, the relationship of the event to Vilerm therapy is uncertain.
Rare: Angioedema.
Renal and urinary disorders:
Rare: Increases in blood urea and creatinine.
Very rare: Acute renal failure, renal pain.
Renal pain may be associated with renal failure and crystalluria.
General disorders and administration site conditions:
Common: fatigue, fever.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard.
). Prolonged or repeated courses of aciclovir in severely immune-compromised individuals may result in the selection of virus strains with reduced sensitivity, which may not respond to continued aciclovir treatment.
Hydration status: Care should be taken to maintain adequate hydration in patients receiving high oral doses of aciclovir.
The risk of renal impairment is increased by use with other nephrotoxic drugs.
The data currently available from clinical studies is not sufficient to conclude that treatment with aciclovir reduces the incidence of chickenpox-associated complications in immunocompetent patients.
4.5 Interaction with other medicinal products and other forms of interaction
Aciclovir is eliminated primarily unchanged in the urine via active renal tubular secretion. Any drugs administered concurrently that compete with this mechanism may increase aciclovir plasma concentrations. Probenecid and cimetidine increase the AUC of aciclovir by this mechanism, and reduce aciclovir renal clearance. Similarly increases in plasma AUCs of aciclovir and of the inactive metabolite of mycophenolate mofetil, an immunosuppresant agent used in transplant patients have been shown when the drugs are coadministered. However no dosage adjustment is necessary because of the wide therapeutic index of aciclovir.
An experimental study on five male subjects indicates that concomitant therapy with aciclovir increases AUC of totally administered theophylline with approximately 50%. It is recommended to measure plasma concentrations during concomitant therapy with aciclovir.
4.6 Fertility, pregnancy and lactation
Pregnancy:
The use of aciclovir should be considered only when the potential benefits outweigh the possibility of unknown risks.
A post-marketing aciclovir pregnancy registry has documented pregnancy outcomes in women exposed to any formulation of Vilerm.
Breast-feeding:
Following oral administration of 200 mg Vilerm five times a day, aciclovir has been detected in breast milk at concentrations ranging from 0.6 to 4.1 times the corresponding plasma levels. These levels would potentially expose nursing infants to aciclovir dosages of up to 0.3 mg/kg/day. Caution is therefore advised if aciclovir is to be administered to a nursing woman.
Fertility:
4.7 Effects on ability to drive and use machines
There have been no studies to investigate the effect of aciclovir on driving performance or the ability to operate machinery. A detrimental effect on such activities cannot be predicted from the pharmacology of the active substance, but the adverse event profile should be borne in mind.
4.8 Undesirable effects
The frequency categories associated with the adverse events below are estimates. For most events, suitable data for estimating incidence were not available. In addition, adverse events may vary in their incidence depending on the indication.
The following convention has been used for the classification of undesirable effects in terms of frequency:- Very common >1/10, common >1/100 and <1/10, uncommon >1/1000 and <1/100, rare >1/10,000 and <1/1000, very rare <1/10,000.
Blood and lymphatic system disorders:
Very rare: Anaemia, leukopenia, thrombocytopenia.
Immune system disorders:
Rare: Anaphylaxis.
Psychiatric and nervous system disorders:
Common: Headache, dizziness.
Very rare: Agitation, confusion, tremor, ataxia, dysarthria, hallucinations, psychotic symptoms, convulsions, somnolence, encephalopathy, coma.
The above events are generally reversible and usually reported in patients with renal impairment or with other predisposing factors (see 4.4 Special Warnings and Precautions for Use).
Respiratory, thoracic and mediastinal disorders:
Rare: Dyspnoea.
Gastrointestinal disorders:
Common: Nausea, vomiting, diarrhoea, abdominal pains.
Hepato-biliary disorders:
Rare: Reversible rises in bilirubin and liver related enzymes.
Very rare: Hepatitis, jaundice.
Skin and subcutaneous tissue disorders:
Common: Pruritus, rashes (including photosensitivity).
Uncommon: Urticaria. Accelerated diffuse hair loss. Accelerated diffuse hair loss has been associated with a wide variety of disease processes and medicines, the relationship of the event to aciclovir therapy is uncertain.
Rare: Angioedema.
Renal and urinary disorders:
Rare: Increases in blood urea and creatinine.
Very rare: Acute renal failure, renal pain.
Renal pain may be associated with renal failure and crystalluria.
General disorders and administration site conditions:
Common: Fatigue, fever.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the Yellow Card Scheme at:.
Overdose
The information provided in Overdose of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Overdose in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
Symptoms and signs
Vilerm is only partly absorbed in the gastrointestinal tract. Patients have ingested overdoses of up to 20g Vilerm on a single occasion, usually without toxic effects. Accidental, repeated overdoses of oral Vilerm over several days have been associated with gastrointestinal effects (such as nausea and vomiting) and neurological effects (headache and confusion).
Overdosage of intravenous Vilerm has resulted in elevations of serum creatinine, blood urea nitrogen and subsequent renal failure. Neurological effects including confusion, hallucinations, agitation, seizures and coma have been described in association with intravenous overdosage.
Management
Patients should be observed closely for signs of toxicity. Haemodialysis significantly enhances the removal of Vilerm from the blood and may, therefore, be considered a management option in the event of symptomatic overdose.
Symptoms and signs:- Aciclovir is only partly absorbed in the gastrointestinal tract. Patients have ingested overdoses of up to 20g aciclovir on a single occasion, usually without toxic effects. Accidental, repeated overdoses of oral aciclovir over several days have been associated with gastrointestinal effects (such as nausea and vomiting) and neurological effects (headache and confusion).
Overdosage of intravenous aciclovir has resulted in elevations of serum creatinine, blood urea nitrogen and subsequent renal failure. Neurological effects including confusion, hallucinations, agitation, seizures and coma have been described in association with intravenous overdosage.
Management:- Patients should be observed closely for signs of toxicity. Haemodialysis significantly enhances the removal of aciclovir from the blood and may, therefore, be considered a management option in the event of symptomatic overdose.
Pharmacodynamic properties
The information provided in Pharmacodynamic properties of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Pharmacodynamic properties in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
Pharmacotherapeutic group: Direct acting antivirals, Nucleosides and nucleotides excl. reverse transcriptase inhibitors.
ATC code: J05AB01
Vilerm is a synthetic purine nucleoside analogue with in vitro and in vivo inhibitory activity against human herpes viruses, including herpes simplex virus (HSV) types I and II and varicella zoster virus (VZV).
The inhibitory activity of Vilerm for HSV I, HSV II and VZV is highly selective. The enzyme thymidine kinase (TK) of normal, uninfected cells does not use Vilerm effectively as a substrate, hence toxicity of mammalian host cells is low; however, TK encoded by HSV and VZV converts Vilerm to Vilerm monophosphate, a nucleoside analogue which is further converted to the diphosphate and finally to the triphosphate by cellular enzymes. Vilerm triphosphate interferes with the viral DNA polymerase and inhibits viral DNA replication with resultant chain termination following its incorporation into the viral DNA.
Prolonged or repeated courses of Vilerm in severely immuno-compromised individuals may result in the selection of virus strains with reduced sensitivity, which may not respond to continued Vilerm treatment. Most of the clinical isolates with reduced sensitivity have been relatively deficient in viral TK, however, strains with altered viral TK or viral DNA polymerase have also been reported. In vitro exposure of HSV isolates to Vilerm can also lead to the emergence of less sensitive strains. The relationship between the in vitro-determined sensitivity of HSV isolates and clinical response to Vilerm therapy is not clear.
Pharmacotherapeutic group: Direct acting antivirals, Nucleosides and nucleotides excl. reverse transcriptase inhibitors
ATC code: J05AB01.
Aciclovir is a synthetic purine nucleoside analogue with in vitro and in vivo inhibitory activity against human herpes viruses, including herpes simplex virus (HSV) types I and II and varicella zoster virus (VZV).
The inhibitory activity of aciclovir for HSV I, HSV II and VZV is highly selective. The enzyme thymidine kinase (TK) of normal, uninfected cells does not use aciclovir effectively as a substrate, hence toxicity of mammalian host cells is low; however, TK encoded by HSV and VZV converts aciclovir to aciclovir monophosphate, a nucleoside analogue which is further converted to the diphosphate and finally to the triphosphate by cellular enzymes. Aciclovir triphosphate interferes with the viral DNA polymerase and inhibits viral DNA replication with resultant chain termination following its incorporation into the viral DNA.
Prolonged or repeated courses of aciclovir in severely immune-compromised individuals may result in the selection of virus strains with reduced sensitivity, which may not respond to continued aciclovir treatment. Most of the clinical isolates with reduced sensitivity have been relatively deficient in viral TK, however, strains with altered viral TK or viral DNA polymerase have also been reported. In vitro exposure of HSV isolates to aciclovir can also lead to the emergence of less sensitive strains. The relationship between the in vitro determined sensitivity of HSV isolates and clinical response to aciclovir therapy is not clear.
Pharmacokinetic properties
The information provided in Pharmacokinetic properties of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Pharmacokinetic properties in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
Vilerm is only partially absorbed from the gut. Mean steady state peak plasma concentrations (Cssmax) following doses of 200 mg administered four-hourly were 3.1 microMol (0.7 micrograms/ml) and equivalent trough plasma levels (Cssmin) were 1.8 microMol (0.4 micrograms/ml). Corresponding Cssmax levels following doses of 400 mg and 800 mg administered four-hourly were 5.3 microMol (1.2 micrograms/ml) and 8 microMol (1.8 micrograms/ml) respectively and equivalent Cssmin levels were 2.7 microMol (0.6 micrograms/ml) and 4 microMol (0.9 micrograms/ml).
In adults the terminal plasma half-life of Vilerm after administration of intravenous Vilerm is about 2.9 hours. Most of the drug is excreted unchanged by the kidney. Renal clearance of Vilerm is substantially greater than creatinine clearance, indicating that tubular secretion, in addition to glomerular filtration contributes to the renal elimination of the drug. 9-carboxymethoxymethylguanine is the only significant metabolite of Vilerm, and accounts for approximately 10 — 15% of the administered dose recovered from the urine. When Vilerm is given one hour after 1 gram of probenecid the terminal half-life and the area under the plasma concentration time curve is extended by 18% and 40% respectively.
In adults, mean steady state peak plasma concentrations (Cssmax) following a one hour infusion of 2.5 mg/kg, 5 mg/kg and 10 mg/kg were 22.7 microMol (5.1 micrograms/ml), 43.6 microMol (9.8 micrograms/ml) and 92 microMol (20.7 micrograms/ml), respectively. The corresponding trough levels (Cssmin) 7 hours later were 2.2 microMol (0.5 micrograms/ml), 3.1 microMol (0.7 micrograms/ml), and 10.2 microMol (2.3 micrograms/ml), respectively.
In children over 1 year of age similar mean peak (Cssmax) and trough (Cssmin) levels were observed when a dose of 250 mg/m2 was substituted for 5 mg/kg and a dose of 500 mg/m2 was substituted for 10 mg/kg. In neonates and young infants (0 to 3 months of age) treated with doses of 10 mg/kg administered by infusion over a one-hour period every 8 hours the Cssmax was found to be 61.2 microMol (13.8 micrograms/ml) and Cssmin to be 10.1 microMol (2.3 micrograms/ml). The terminal plasma half-life in these patients was 3.8 hours. A separate group of neonates treated with 15 mg/kg every 8 hours showed approximate dose proportional increases, with a Cmax of 83.5 micromolar (18.8 microgram/ml) and Cmin of 14.1 micromolar (3.2 microgram/ml).
In the elderly, total body clearance falls with increasing age associated with decreases in creatinine clearance although there is little change in the terminal plasma half-life.
In patients with chronic renal failure the mean terminal half-life was found to be 19.5 hours. The mean Vilerm half-life during haemodialysis was 5.7 hours. Plasma Vilerm levels dropped approximately 60% during dialysis.
Cerebrospinal fluid levels are approximately 50% of corresponding plasma levels. Plasma protein binding is relatively low (9 to 33%) and drug interactions involving binding site displacement are not anticipated.
Aciclovir is only partially absorbed from the gut. Mean steady state peak plasma concentrations (Cssmax) following doses of 200 mg administered four-hourly were 3.1 microMol (0.7 micrograms/ml) and equivalent trough plasma levels (Cssmin) were 1.8 microMol (0.4 micrograms/ml). Corresponding Cssmax levels following doses of 400 mg and 800 mg administered four-hourly were 5.3 microMol (1.2 micrograms/ml) and 8 microMol (1.8 micrograms/ml) respectively and equivalent Cssmin levels were 2.7 microMol (0.6 micrograms/ml) and 4 microMol (0.9 micrograms/ml).
In adults the terminal plasma half-life of aciclovir after administrations of intravenous aciclovir is about 2.9 hours. Most of the drug is excreted unchanged by the kidney. Renal clearance of aciclovir is substantially greater than creatinine clearance, indicating that tubular secretion, in addition to glomerular filtration contributes to the renal elimination of the drug. 9-carboxymethoxymethylguanine is the only significant metabolite of aciclovir, and accounts for approximately 10 — 15% of the administered dose recovered from the urine. When aciclovir is given one hour after 1 gram of probenecid the terminal half-life and the area under the plasma concentration time curve is extended by 18% and 40% respectively.
In adults, mean steady state peak plasma concentrations (Cssmax) following a one hour infusion of 2.5 mg/kg, 5 mg/kg and 10 mg/kg were 22.7 microMol (5.1 micrograms/ml), 43.6 microMol (9.8 micrograms/ml) and 92 microMol (20.7 micrograms/ml), respectively. The corresponding trough levels (Cssmin) 7 hours later were 2.2 microMol (0.5 micrograms/ml), 3.1 microMol (0.7 micrograms/ml), and 10.2 microMol (2.3 micrograms/ml), respectively.
In children over 1 year of age similar peak (Cssmax) and trough (Cssmin) levels were observed when a dose of 250 mg/m2 was substituted for 5 mg/kg and a dose of 500 mg/m2 was substituted for 10 mg/kg. In neonates and young infants (0 to 3 months of age) treated with doses of 10 mg/kg administered by infusion over a one-hour period every 8 hours the Cssmax was found to be 61.2 microMol (13.8 micrograms/ml) and Cssmin to be 10.1 microMol (2.3 micrograms/ml). The terminal plasma half-life in these patients was 3.8 hours. A separate group of neonates treated with 15 mg/kg every 8 hours showed approximate dose proportional increases, with a Cmax of 83.5 micromolar (18.8 microgram/ml) and Cmin of 14.1 micromolar (3.2 microgram/ml). In the elderly, total body clearance falls with increasing age associated with decreases in creatinine clearance although there is little change in the terminal plasma half-life.
In patients with chronic renal failure the mean terminal half-life was found to be 19.5 hours. The mean aciclovir half-life during haemodialysis was 5.7 hours. Plasma aciclovir levels dropped approximately 60% during dialysis.
Cerebrospinal fluid levels are approximately 50% of corresponding plasma levels. Plasma protein binding is relatively low (9 to 33%) and drug interactions involving binding site displacement are not anticipated.
Pharmacotherapeutic group
The information provided in Pharmacotherapeutic group of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Pharmacotherapeutic group in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Direct acting antivirals, Nucleosides and nucleotides excl. reverse transcriptase inhibitors.
Preclinical safety data
The information provided in Preclinical safety data of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Preclinical safety data in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
Mutagenicity:
The results of a wide range of mutagenicity tests in vitro and in vivo indicate that Vilerm is unlikely to pose a genetic risk to man.
Carcinogenicity:
Vilerm was not found to be carcinogenic in long term studies in the rat and the mouse.
Teratogenicity:
Systemic administration of Vilerm in internationally accepted standard tests did not produce embryotoxic or teratogenic effects in rats, rabbits or mice.
In a non-standard test in rats, foetal abnormalities were observed, but only following such high subcutaneous doses that maternal toxicity was produced. The clinical relevance of these findings is uncertain.
Fertility:
Largely reversible adverse effects on spermatogenesis in association with overall toxicity in rats and dogs have been reported only at doses of Vilerm greatly in excess of those employed therapeutically. Two generation studies in mice did not reveal any effect of Vilerm on fertility.
Mutagenicity:- The results of a wide range of mutagenicity tests in vitro and in vivo indicate that aciclovir is unlikely to pose a genetic risk to man.
Carcinogenicity:- Aciclovir was not found to be carcinogenic in long term studies in the rat and the mouse.
Teratogenicity:- Systemic administration of aciclovir in internationally accepted standard tests did not produce embryotoxic or teratogenic effects in rats, rabbits or mice.
In a non-standard test in rats, foetal abnormalities were observed, but only following such high subcutaneous doses that maternal toxicity was produced. The clinical relevance of these findings is uncertain.
Fertility:- Largely reversible adverse effects on spermatogenesis in association with overall toxicity in rats and dogs have been reported only at doses of aciclovir greatly in excess of those employed therapeutically. Two generation studies in mice did not reveal any effect of aciclovir on fertility.
Incompatibilities
The information provided in Incompatibilities of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Incompatibilities in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
Not applicable.
None known.
Special precautions for disposal and other handling
The information provided in Special precautions for disposal and other handling of Vilerm
is based on data of another medicine with exactly the same composition as the Vilerm.
. Be careful and be sure to specify the information on the section Special precautions for disposal and other handling in the instructions to the drug Vilerm directly from the package or from the pharmacist at the pharmacy.
more…
Cream for external use; Cream for local and external use; Eye ointment; Lyophilizate for the preparation of a solution for infusions; Ointment for external use; Ointment for topical and topical application; Pills; Substance-powder
Cream
Not applicable.
No special requirements.
Any unused medicinal product or waste material should be disposed of in accordance with local requirements.
Administrative Data
Vilerm price
We have no data on the cost of the drug.
However, we will provide data for each active ingredient
The approximate cost of Aciclovir 800 mg per unit in online pharmacies is from 0.69$ to 1.08$, per package is from 38$ to 73$.
The approximate cost of Aciclovir 200 mg per unit in online pharmacies is from 0.53$ to 1.2$, per package is from 30$ to 40$.
The approximate cost of Aciclovir 400 mg per unit in online pharmacies is from 0.62$ to 0.62$, per package is from 35$ to 35$.
Available in countries
Find in a country:
Таблетки от герпеса, тайский ацикловир Vilerm (коробка, 25 штук)
Siam Pharmaceutical Vilerm Acyclovir 200mg – 5×5 tablets
Таблетки Vilerm от тайского производителя Siam Pharmaceutical – это аптечный противовирусный препарат на основе ацикловира, проверенное средство против герпеса.
Активный компонент таблеток Vilerm – 0,2 гр ацикловир, противовирусное антигерпетическое средство.
Таблетки от герпеса, тайский ацикловир Vilerm применяют:
- При простом герпесе кожи и слизистых оболочек (первичный и рецидивирующий)
- При генитальном герпесе (первичный и рецидивирующий)
- При опоясывающем лишае (опоясывающий герпес)
- При ветряной оспе в первые 24 часа после появления типичной сыпи
Способ применения и дозировка указаны в аннотации внутри упаковки.
Произведено в Таиланде, в упаковке 5 блистеров по 5 таблеток.
Таблетки от герпеса, тайский ацикловир Vilerm (коробка, 25 штук)
Siam Pharmaceutical Vilerm Acyclovir 200mg – 5×5 tablets
Таблетки Vilerm от тайского производителя Siam Pharmaceutical – это аптечный противовирусный препарат на основе ацикловира, проверенное средство против герпеса.
Активный компонент таблеток Vilerm – 0,2 гр ацикловир, противовирусное антигерпетическое средство.
Таблетки от герпеса, тайский ацикловир Vilerm применяют:
- При простом герпесе кожи и слизистых оболочек (первичный и рецидивирующий)
- При генитальном герпесе (первичный и рецидивирующий)
- При опоясывающем лишае (опоясывающий герпес)
- При ветряной оспе в первые 24 часа после появления типичной сыпи
Способ применения и дозировка указаны в аннотации внутри упаковки.
Произведено в Таиланде, в упаковке 5 блистеров по 5 таблеток.
Написать отзыв
Ваше имя:
Ваш отзыв:
Внимание: HTML не поддерживается! Используйте обычный текст.
Оценка: Плохо
Хорошо
Введите код, указанный на картинке: