Онсиор™ таблетки инструкция по применению
📜 Инструкция по применению Онсиор™ таблетки
💊 Состав препарата Онсиор™ таблетки
✅ Применение препарата Онсиор™ таблетки
📅 Условия хранения Онсиор™ таблетки
⏳ Срок годности Онсиор™ таблетки
Описание лекарственного препарата ветеринарного назначения Онсиор™ таблетки
Основано на официально утвержденной инструкции по применению
препарата Онсиор™ таблетки для специалистов
и утверждено компанией-производителем для электронного издания справочника Видаль Ветеринар
2019 года
Дата обновления: 2018.05.21
Лекарственная форма
|
|
Онсиор™ таблетки |
Таблетки для орального применения (для кошек) 6 мг рег. 826-3-2.17-3579№ПВИ-3-2.17/04923 |
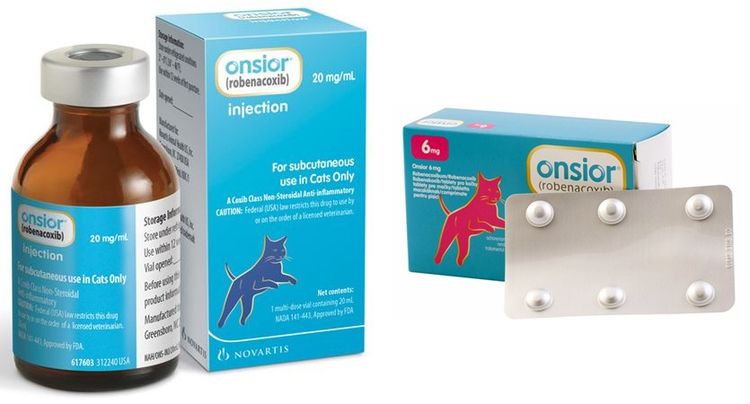
Форма выпуска, состав и упаковка
Таблетки для орального применения (для кошек) от бежевого до коричневого цвета, круглые, с оттиском «NA» на одной стороне и оттиском «АК» на другой стороне.
Вспомогательные вещества: дрожжи, целлюлоза микрокристаллическая, повидон (К-30), кросповидон, кремния диоксид коллоидный безводный, магния стеарат.
Расфасованы по 6 таблеток в блистеры из алюминиевой фольги, помещенные по 1, 2, 5 или 10 шт. в картонные пачки вместе с инструкцией по применению.
Фармакологические (биологические) свойства и эффекты
Робенакоксиб — НПВС группы коксибов, является селективным специфическим ингибитором ЦОГ-2 и обладает противовоспалительным и анальгезирующим действием. ЦОГ представлена в двух формах: ЦОГ-1 — конститутивная форма энзима, выполняет защитные функции, в т.ч. в ЖКТ и почках; ЦОГ-2 — индуцируемая форма энзима, отвечает за выработку медиаторов, включающих ПГЕ2, вызывающих боль, воспаление и жар.
После подкожного введения препарата робенакоксиб быстро всасывается из места инъекции, при пероральном введении — быстро всасывается в кишечнике. Поступает в системный кровоток и большинство органов и тканей; более 99% робенакоксиба связывается с белками плазмы. Максимальная концентрация в плазме при применении раствора для инъекций отмечается через 1 ч, биодоступность составляет 69% у кошек и 88% у собак. При пероральном применении максимальная концентрация в плазме отмечается через 0.5 ч, биодоступность составляет 49% у кошек и 62% у собак при применении таблеток с кормом и 84% — без корма.
Метаболизируется робенакоксиб в печени, выделяется в основном с желчью (около 70% у кошек и около 65% у собак) и частично — с мочой; период полувыведения из организма собак составляет около 1.2 ч, кошек — 1.1-1.7 ч.
Онсиор™ по степени воздействия на организм относится к умеренно опасным веществам (3 класс опасности по ГОСТ 12.1.007).
Показания к применению препарата ОНСИОР™
Назначают собакам и кошкам в качестве противовоспалительного и болеутоляющего препарата при воспалительных и болевых синдромах различного происхождения, в т.ч.:
- острые и хронические заболевания опорно-двигательного аппарата (артриты, артрозы, синовиты, вывихи);
- воспалительные заболевания мягких тканей;
- в качестве анальгезирующего и противовоспалительного препарата в послеоперационный период.
Порядок применения
Раствор для инъекций
Применяют кошкам и собакам в дозе 1 мл на 10 кг массы животного (что соответствует 2 мг робенакоксиба на 1 кг массы тела животного).
При хирургических вмешательствах препарат вводят примерно за 30 мин до введения в наркоз. В послеоперационный период в качестве анальгезирующего и противовоспалительного препарат вводят в течение 1-2 дней.
При лечении заболеваний опорно-двигательного аппарата препарат применяют для снятия острой боли однократно или двукратно в дозе 1 мл на 10 кг массы животного (что соответствует 2 мг робенакоксиба/кг массы животного); дальнейшее лечение рекомендуется продолжить препаратом в форме таблеток в соответствии с инструкцией по применению.
Таблетки
Применяют собакам и кошкам индивидуально, 1 раз/сут, в дозах, указанных в таблице:
Рекомендуется давать препарат отдельно от кормления, но, при необходимости, препарат может быть дан вместе с кормом. Таблетки не следует дробить или разламывать.
Онсиор™ таблетки для кошек применяют при оперативных вмешательствах за 30 мин до введения в наркоз в дозе 1-2.4 мг/кг массы животного; лечение можно продолжать в течение периода длительностью до 2 дней после операции; при заболеваниях опорно-двигательного аппарата препарат применяют до 6 дней.
Онсиор™ таблетки для собак применяют за 30 мин до или после кормления в дозе 1-2 мг/кг массы животного.
Курс лечения определяет лечащий ветеринарный врач. Как правило, продолжительность лечения составляет 7 дней. Если по истечении 10 дней клиническое улучшение не наступает, применение препарата прекращают.
Особенностей действия лекарственного препарата при его первом применении и отмене не установлено.
Следует избегать нарушения рекомендуемого интервала между введениями препарата, т.к. это может привести к снижению терапевтической эффективности. В случае пропуска повторного введения препарат следует ввести как можно скорее в предусмотренной дозе.
Побочные эффекты
При применении препарата в соответствии с инструкцией побочных явлений и осложнений у животных, как правило, не наблюдается. У кошек и собак возможны желудочно-кишечные расстройства (рвота, мягкий кал или диарея), указанные симптомы проходят самопроизвольно и не требуют применения лекарственных средств. При индивидуальной повышенной чувствительности животного к препарату и появлении аллергических реакций, применение препарата прекращают и животному назначают антигистаминные и симптоматические средства.
При передозировке у животных с выраженной чувствительностью к компонентам препарата могут наблюдаться расстройство ЖКТ, печеночная или почечная недостаточность. Специфические средства детоксикации отсутствуют, применяют общие меры, направленные на выведение лекарственного препарата из организма.
Противопоказания к применению препарата ОНСИОР™
- индивидуальная повышенная чувствительность животного к компонентам препарата;
- гиповолемия;
- артериальная гипотензия;
- состояние дегидратации;
- масса животного менее 2.5 кг.
При крайней необходимости назначения лекарственного препарата таким животным лечение проводят под постоянным наблюдением ветеринарного врача.
Не следует применять препарат животным с выраженной сердечной, почечной и печеночной недостаточностью.
Особые указания и меры личной профилактики
Запрещено применять препарат беременным и лактирующим самкам, щенкам моложе 2-месячного и котятам моложе 4-месячного возраста.
Препарат не предназначен для применения продуктивным животным.
Онсиор™ раствор для инъекций не следует применять одновременно с другими НПВС, а также смешивать в одном шприце с другими лекарственными препаратами.
Онсиор™ таблетки не следует применять одновременно с другими НПВС и в течение 24 ч после их отмены, диуретиками, а также ингибиторами АПФ и другими лекарственными средствами с высокой степенью связывания с белками сыворотки крови из-за вероятности взаимного усиления токсического действия.
Меры личной профилактики
При применении препарата следует соблюдать общие правила личной гигиены и техники безопасности, предусмотренные при работе с лекарственными препаратами. Во время работы запрещается курить, пить и принимать пищу. По окончании работы следует тщательно вымыть с мылом лицо и руки.
Людям с гиперчувствительностью к компонентам препарата следует избегать прямого контакта с препаратом Онсиор™. При случайном контакте лекарственного препарата с кожей или слизистыми оболочками их необходимо немедленно промыть большим количеством воды. В случае появления аллергических реакций или при случайном попадании препарата в организм человека следует немедленно обратиться в медицинское учреждение (при себе иметь инструкцию по применению препарата или этикетку).
Запрещается использование пустой упаковки из-под препарата для бытовых целей; она подлежит утилизации с бытовыми отходами.
Условия хранения Онсиор™ таблетки
Онсиор™ раствор для инъекций следует хранить в закрытой упаковке производителя, отдельно от пищевых продуктов и кормов, в защищенном от прямых солнечных лучей, недоступном для детей месте при температуре от 2°С до 8°С.
Онсиор™ таблетки следует хранить в закрытой упаковке производителя, отдельно от пищевых продуктов и кормов, в защищенном от прямых солнечных лучей, недоступном для детей месте при температуре от 0°С до 25°С. Срок годности при соблюдении условий хранения — 4 года с даты производства.
Запрещается применение препарата по истечении срока годности.
Неиспользованный лекарственный препарат утилизируют в соответствии с требованиями законодательства.
Срок годности Онсиор™ таблетки
Срок годности при соблюдении условий хранения — 3 года с даты производства, после первого вскрытия флакона — 28 суток.
Условия отпуска
Препарат отпускается без рецепта.
Контакты для обращений
|
|
123112 Москва, Пресненская наб. 10 |
Онсиор™ таблетки отзывы
Помогите другим с выбором, оставьте отзыв об Онсиор™ таблетки
Имя: Ольга
Дата: 2022-12-03
Очень помогли моему коту.который уже не вставал только лежал
Оставить отзыв
Если вы хотите разместить ссылку на описание этого препарата — используйте данный код

(robenacoxib)
6 mg tablets for cats
Non-Steroidal Anti-Inflammatory
For Veterinary Use Only
For Use in Cats Only
DIN 02375834
Description
ONSIOR contains robenacoxib which is a non-steroidal anti-inflammatory drug (NSAID) belonging to the coxib class. The flavoured tablets are round, beige to brown in colour and are not scored.
Onsior (tablets for cats) (6 mg) Indications
ONSIOR tablets are indicated:
1. for the relief of acute pain and inflammation associated with cat bites and scratches with and without abscesses and musculoskeletal injuries such as sprains and strains in cats.
2. as an adjunctive medication, in the control of postoperative pain and inflammation associated with onychectomy, ovariohysterectomy, and castration in cats.
3. for the control of pain and inflammation associated with chronic musculoskeletal disorders, such as osteoarthritis, in cats.
Dosage and Administration
For oral use in cats ≥ 2.5 kg AND ≥ 4 months of age.
Carefully consider the potential benefits and risks of ONSIOR and other treatment options before deciding to use ONSIOR (robenacoxib). Use the lowest effective dose for the shortest duration consistent with individual response.
Give orally without food or with a small quantity of food (see PHARMACOLOGY).
The recommended daily dose of ONSIOR is 1 mg/kg body weight with a range 1 to 2.4 mg/kg (see Dosing Chart below).
For relief of acute pain and inflammation associated with cat bites and scratches with and without abscesses and musculoskeletal injuries, administer once per day for a maximum of 6 days according to the dosing chart below.
As an adjunctive medication, in the control of postoperative pain and inflammation associated with onychectomy, ovariohysterectomy and castration, administer once per day for a maximum of 3 days according to the dosing chart below. The first dose should be administered approximately 30 minutes (without food) before the start of surgery, around the time of induction of general anesthesia.
After surgery, once daily treatment may be continued for up to two days with either ONSIOR tablets or ONSIOR injection at their respective label recommended dose. If subsequent doses are given by subcutaneous injection, different sites for each injection should be used. If necessary, additional analgesic treatment with opioids is recommended.
For the control of pain and inflammation associated with chronic musculoskeletal disorders, such as osteoarthritis, in cats, administer once daily according to the table below. Duration of treatment should be decided on an individual basis. Response to treatment should be monitored at regular intervals by the responsible veterinarian. In clinical field studies, robenacoxib was well-tolerated by most cats for periods up to 12 weeks. A clinical response is normally seen within 3 to 6 weeks. Treatment should be discontinued after 6 weeks if no clinical improvement is apparent.
Note: In cats, the dose of ONSIOR tablets and ONSIOR injectable are different.
Dosing Chart:
|
Body Weight |
ONSIOR (robenacoxib) 6 mg Tablets / Frequency |
|
2.5 to less than 6 kg |
1 whole tablet once daily |
|
6 to less than 12 kg |
2 whole tablets once daily |
Tablets are not scored and should not be broken.
Do not use in cats weighing less than 2.5 kg as these animals cannot be accurately dosed.
As with other NSAIDs, do not exceed the recommended dosage (see CONTRAINDICATIONS and CAUTIONS sections). When prescribing NSAIDs, exceeding the label recommended dose could lead to complications, including acute renal failure and in some cases associated death has been reported.
Owners should be advised when their pet has received a robenacoxib injection prior to initiating the use of ONSIOR tablets, and be informed of the potential for adverse reactions and clinical signs associated with possible NSAID intolerance. Always provide the Client Information Sheet with prescription (detach from Package Insert).
Contraindications
As with all non-steroidal anti-inflammatory drugs (NSAIDs), administration of this drug is contraindicated in the following circumstances:
● cats with gastrointestinal ulcers;
● cats that are dehydrated, hypovolemic, hypoproteinemic or hypotensive;
● cats with impaired cardiac, renal or hepatic function, or coagulation abnormalities;
● a known hypersensitivity to robenacoxib or its excipients;
● concurrent use of other NSAIDs or corticosteroids;
● in breeding, pregnant or lactating cats because the safety of robenacoxib has not been established in these animals.
CAUTIONS:
The safety of ONSIOR tablets has not been established in cats weighing less than 2.5 kg or under 4 months of age.
Prolonged treatment with robenacoxib for the relief of chronic pain and inflammation should only be considered after a risk/benefit assessment by the responsible veterinarian.
All cats should undergo a thorough history and physical exam before the initiation of NSAID therapy. Appropriate laboratory tests should be conducted to establish hematological and biochemical baseline data before the administration of any NSAID. ONSIOR should be used with caution in cats with a known hypersensitivity to other NSAIDs.
As a class, cyclo-oxygenase inhibitory NSAIDs may be associated with gastrointestinal, renal, and hepatic toxicity. Sensitivity to drug-associated adverse events varies with the individual patient. Cats that have experienced adverse reactions from one NSAID may experience adverse reactions from another NSAID. Patients at greatest risk for adverse events are those that are dehydrated, on concomitant diuretic therapy, or those with existing renal, cardiovascular, and/or hepatic dysfunction. Anesthetic drugs may affect renal perfusion; approach concomitant use of anesthetics and NSAIDs cautiously. Appropriate monitoring procedures (including ECG, blood pressure, and temperature regulation) should be employed during all surgical procedures. The use of parenteral fluids during surgery is recommended to decrease potential renal complications when using NSAIDs perioperatively.
If decrease in appetite, vomiting, diarrhea, change in drinking or urination, change in behaviour, such as depression or restlessness, or other suspected adverse reactions occur, stop administration of ONSIOR immediately and seek the advice of a veterinarian (see ADVERSE REACTIONS).
Pre-treatment with other anti-inflammatory drugs may result in additional or increased adverse effects and accordingly a treatment-free period with such substances should be observed. The treatment-free period should take into account the pharmacokinetic properties of the products used previously.
Concomitant treatment with drugs which affect renal flow, e.g. anesthetics, should be subject to clinical monitoring. Concurrent administration of potentially nephrotoxic medicines should be avoided as there might be an increase of renal toxicity. The use of concomitantly protein-bound drugs with ONSIOR Tablets for Cats has not been studied in cats. Drug compatibility should be monitored closely in patients requiring additional therapy.
ONSIOR was administered concurrently with pre-anesthetic (opioid) and a metacarpal four point ring block using a longer acting local anesthetic medication, during the field study assessing the effect of ONSIOR on postoperative pain control. Level of analgesia should be monitored postoperatively to assess if additional pain control is needed. If necessary, additional analgesic treatment with opioids is recommended. Other NSAIDs and corticosteroids should not be used.
The use of ONSIOR in cats with cardiac disease has not been studied. ONSIOR has been shown to prolong the QT interval in repeat dose laboratory studies. In anaesthetised healthy cats, a single intravenous administration of 2 or 4 mg/kg ONSIOR had no effect on the ECG including the QT interval. The associated risk of developing a ventricular arrhythmia is unknown. The use of robenacoxib with other drugs shown to prolong the QT interval is not recommended. Commonly used drugs that prolong QT interval include antihistamines and prokinetic drugs. Appropriate monitoring procedures including ECG should be employed during all surgical procedures.
Studies to determine the activity of robenacoxib when administered concomitantly with other protein-bound or similarly metabolized drugs have not been conducted. Commonly used protein-bound drugs include cardiac, anticonvulsant, and behavioural medications. The influence of concomitant drugs that may inhibit metabolism of ONSIOR has not been evaluated. Drug compatibility should be monitored in patients requiring adjunctive therapy.
A gamma lactam (γ-lactam) metabolite of robenacoxib is formed in the cat. In addition, this γ-lactam is a degradation product that increases over the shelf life of the tablets and can be decreased by refrigeration. Neurological signs have been associated with the use of β lactam drugs; it is unknown if this γ-lactam may cause similar neurological signs.
Warnings
KEEP OUT OF REACH OF CHILDREN. In case of accidental ingestion, seek medical advice immediately and show the package insert or the label to the physician.
Adverse Reactions
Although all adverse events are not reported, the following information is based on voluntary post-approval drug experience reporting. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using this data.
The post-marketing adverse event reports for ONSIOR 6 mg tablets for cats have shown the following signs to be reported very rarely (less than 1 animal in 10,000 animals, including isolated reports): anorexia, lethargy, vomiting, and diarrhea, elevated creatinine, elevated BUN, and renal insufficiency.
For technical support or to report a suspected adverse drug reaction, contact Elanco Canada Limited at 1-800-265-5475.
Information for Cat Owners:
ONSIOR is the product your veterinarian has chosen to treat your cat’s pain and inflammation. It belongs to a class of drugs called “non-steroidal anti-inflammatory” or NSAIDs. These drugs must be used according to your veterinarian’s directions. Occasionally, NSAIDs can cause side effects. If your cat stops eating, is depressed, vomits or has diarrhea stop the drug immediately and contact your veterinarian. In most cases the side effects will disappear when the drug is stopped but in rare cases it may be serious. For this reason, it is important to consult with your veterinarian. If you notice any serious side effects or others not mentioned in the insert, please inform your veterinarian.
Pharmacology
Mode of Action
Pharmacodynamics
Robenacoxib is an NSAID of the coxib class. It is a highly potent and selective inhibitor of the cyclooxygenase 2 enzyme (COX-2). The cyclooxygenase enzyme (COX) is present in two forms. COX-1 is the “constitutive” form of the enzyme and has protective functions including in the gastrointestinal tract and kidney. COX-2 is the “inducible” form of the enzyme and is responsible for the production of mediators including PGE2 which induce pain, inflammation or fever.
Pharmacokinetics
Absorption: In a feline pharmacokinetic study, estimates for PK parameters in fasted cats included Cmax 1158.9 ng/mL, Tmax 0.5 h, AUC (0-∞) 1337 ng.h/mL. Co-administration with the normal diet (1/3 of daily ration) produced no change in Cmax, Tmax or AUC (0-∞). Co-administration with the entire daily ration of food produced no change in Tmax but a 20% reduction in AUC (0-∞) and a 40.3% reduction in Cmax of robenacoxib.
The systemic bioavailability of non-flavoured robenacoxib tablets was 49% without food.
Distribution: Robenacoxib has a relatively small volume of distribution (Vss 190 mL/kg) and is highly bound to plasma proteins (>98%).
Biotransformation: Robenacoxib is extensively metabolized by the liver in cats. The systemic exposure of the γ-lactam metabolite is about 25% of robenacoxib exposure following oral administration to fed cats. Further, the systemic exposure to lactam appears to be two-fold greater in fed cats than in fasted cats (see CAUTIONS). Apart from the one γ-lactam metabolite, the identity and activity of the other metabolites is not known in cats.
Elimination: Robenacoxib is rapidly cleared from blood (CL 0.44 L/kg/h) with a mean elimination half-life 1.1 h after intravenous administration. After oral administration of tablets, the mean terminal half-life from blood was 1.7 h, and elimination is predominantly through the biliary route (fecal and urinary excretion are 60 and 16.5% respectively).
The pharmacokinetics of robenacoxib do not differ between male and female cats.
SAFETY STUDIES:
21-Day Target Animal Safety Study:
In a 21-day laboratory tolerance study, 8 month-old healthy, DSH cats (4/sex/group) were administered robenacoxib at a dose of 0 mg/kg (control group) or 24 mg/kg/day (10X the maximum exposure based on the single, 6 mg tablet size). All cats survived to study termination. Vomiting and decreased activity was noted in some of the treated cats. Two cats in the 10X group exhibited abnormal rear limb neurologic function. One of these cats also exhibited a head tilt and nystagmus at the end of the study. Mean food consumption was less in the 10X group. The mean kidney weights were lower in the 10X group compared to the control group; and the mean thymus weights were also lower in the 10X group compared to the controls. Two cats in the 10X group had chronic interstitial nephritis on histopathology; this finding was correlated with a renal mass observed on gross necropsy in one cat. One 10X cat had a focal cecal/large intestinal erosion. One 10X cat and one control cat had periportal, multifocal necrosis in one lobe of the liver. There were four 10X cats and 2 control cats with renal tubular degeneration. Under the conditions of this study, robenacoxib was well tolerated when administered at 24 mg/kg/day for 21 days, except for 2 cats in the 10X group with neurologic signs.
42-Day Target Animal Safety Study:
In a 42-day study, 8 month-old, healthy cats were administered robenacoxib at 0, 2, 6 or 10 mg/kg/twice daily. Small thymuses were noted in all robenacoxib-treated groups with corresponding organ weight decreases and/or atrophic changes on histopathology. There was a decrease in the kidney weights in the 10 mg/kg/twice daily-group compared to the controls. Vomiting was the most common adverse reaction noted in the treated cats. An adequate safety margin was demonstrated for ONSIOR when administered under the conditions of this 42-day study.
6 Month Target Animal Safety Study:
In a 6 month study, 8 month old, healthy cats (4/sex/group) were administered robenacoxib at 0, 1X (2.4 mg/kg), 3X (7.2 mg/kg) or 5X (12 mg/kg) once daily. One 5X cat had clonic seizures on Day 115 and was ataxic on Day 175. One 5X cat had skin cold to the touch on Day 106. One cat in the 1X group experienced urethral obstruction/FLUTD. Vomiting, decreased activity, injected sclera and soft stools were the most common adverse reactions observed in the treated groups. Soft stools and injected sclera were also observed in the control group.
The mean body weights in the 1X and 3X groups were lower than the controls from Day 21 to Day 182; and lower in the 5X group from Day 28 to Day 182. There was a clear dose-related and possibly time-related increase in the QTc interval at Day 41 and Day 175, particularly in the 3X and 5X groups, no other ECG abnormalities were noted.
There was no obvious accumulation in Cmax or AUC between Days 1, 31 and 171, and there was no apparent difference in parameters between males and females. The following parameters were calculated for the 1X dosage: Tmax was 0.5 h (median), the dose-normalized mean Cmax was 668 ng/mL and the dose-normalized mean area under the curve (AUC(0-inf)) was 902 h*ng/mL. Similarly, the following parameters were calculated for the 3X dosage: Tmax was 0.5 h (median), the dose normalized mean Cmax was 1019 ng/mL and the dose-normalized mean area under the curve (AUC(0-inf)) was 1394 h*ng/mL. For the 5X dosage the following parameters were calculated: Tmax was 1.0 h (median), the dose-normalized mean Cmax was 1198 ng/mL and the dose-normalized mean area under the curve (AUC(0-inf)) was 1884 h*ng/mL. A post hoc analysis of PK parameters revealed that dose normalized Cmax and AUC were greater than dose proportional.
The mean kidney weights were lower in all robenacoxib-treated groups. One 5X cat with decreased kidney weight and size also had transient increases in BUN and creatinine. There were transient increases in AST, amylase, and ALT in the 3X and 5X cats from Day 30 to Day 183. There was an increased severity of the tubular degeneration/regeneration in the kidneys of two 1X and two 5X cats with inflammation, papillary necrosis and papillary mineralization. An increased incidence of minimal to mild Kupffer cell pigmentation was observed in the livers of all robenacoxib-treated cats; however, no hepatocellular damage was noted on histopathology. One 5X cat had a focal, minimal ulcer of the gastric fundus (peptic and parietal cells).
This 6 month safety study supports the safe use of ONSIOR for the relief of acute pain and inflammation associated with cat bites and scratches with and without abscesses and musculoskeletal injuries such as sprains and strains in cats for a maximum of 6 days.
37-Day Interchangeable Use Study:
ONSIOR was administered orally (6 mg tablets) and subcutaneously (20 mg/mL solution) to 4-month old healthy cats at 0, 1, 2, and 3 times the labeled doses (1X = 2.4 mg/kg/day orally based on the inherent tablet dose band or 2 mg/kg/day subcutaneously). Interchangeable use was evaluated by alternating three 7-day oral tablet/3-day subcutaneous injection cycles followed by one final 7-day oral tablet dosing cycle. Findings included: elevated creatine kinase levels on Days 13 and 37, soft stools, histologic observation of a minimal oral (tongue) ulceration in a 1X cat, injection site edema for up to 120 h prior to resolution, and a prolonged QT interval in treated cats as compared to the controls on Day 36. Histologically, the injection site had minimal or mild, subacute/chronic inflammation. Inflammation at the injection site was observed in both treated and control animals with a greater frequency in the higher dose groups than in the control and 1X groups. One male and one female in the 1x group had focal tubular degeneration/regeneration of the renal cortex. Focal tubular degeneration/regeneration represents non-specific renal findings which can occur as a result of many causes. Dose-normalized AUC and concentration levels were higher following the oral route than the subcutaneous route. There was no significant accumulation following once daily administration. One 2X-treated cat had a 7-fold increase in buccal mucosal bleeding time (BMBT) during the treatment period compared to the pre-treatment value.
Preliminary 37-Day Interchangeable Use Study:
ONSIOR was administered orally (6 mg tablets) and subcutaneously (20 mg/mL solution) to 4-month old cats at 0, 1, and 5 times the labeled doses (1X = 2.4 mg/kg/day orally based on the inherent tablet dose band or 2 mg/kg/day subcutaneously). Interchangeable use was evaluated by alternating three 7-day oral tablet/3-day subcutaneous injection cycles followed by one final 7-day oral tablet dosing cycle. Clinical findings included: scabs and sores at the injection sites of one 1X female and two 5X females, and injection site edema noted more frequently in treated cats. Injection site changes were characterized as minimal to moderate granulomatous inflammation, minimal to moderate fibroplasia/fibrosis, and minimal myofiber regeneration of the panniculus carnosus. In one 1X female, moderate necrosis of a blood vessel was noted within the granulomatous inflammation. Minimal myofiber regeneration was observed in the underlying skeletal muscle in three out of four 5X males.
A red depressed area on the upper lip of one 5X cat correlated histologically with a minimal ulcer. Creatinine was significantly increased in 5X cats compared to the controls. Urine specific gravities remained within normal limits for all 5X cats, and blood urea nitrogen (BUN) values remained within normal limits for all study animals. Histologically, renal changes included bilateral or unilateral minimal to moderate vacuolation and bilateral or unilateral minimal to mild degeneration of proximal tubules were observed in three 5X males. Two 5X males had mineralized foci in the epithelium covering the papilla.
One 5X female had a brief episode of ataxia and lethargy on Day 16. This cat was subsequently noted to be dehydrated and constipated, requiring veterinary intervention with subcutaneous fluid therapy and nutritional supplementation. This cat had the greatest QT increase on ECG evaluation.
Clinical Safety
In the pivotal clinical trial to evaluate acute pain and inflammation associated with musculoskeletal disorders, where 56 cats were treated with ONSIOR, diarrhea, emesis, lethargy, hyperactivity and polydipsia were reported at the following frequencies:
Adverse Reactions in the Acute Musculoskeletal Disorder Field Study
|
Adverse Reactions |
ONSIOR 6 mg Tablets |
Positive Control |
|
Diarrhea |
4 (7%) |
1 (2%) |
|
Emesis |
3 (5%) |
0 (0%) |
|
Lethargy |
3 (5%) |
1 (2%) |
|
Hyperactivity |
2 (4%) |
0 (0%) |
|
Polydipsia |
1 (2%) |
0 (0%) |
In a placebo controlled postoperative pain field study associated with ovariohysterectomy, castration and onychectomy, 249 male and female cats of various breeds, 6 months to 13 years old weighing 2.5 to 7.5 kg, the most commonly reported adverse reactions were surgical site bleeding, infected surgery sites, lethargy, vomiting and inappetence.
Adverse Reactions in the Postoperative Pain Field Study
|
Clinical Sign* |
ONSIOR 6 mg Tablets (n=167 cats) |
Positive Control (n=82 cats) |
|
Incision site bleeding |
7 (4%) |
1 (1%) |
|
Incision site infection |
6 (4%) |
2 (2%) |
|
Inappetence, weight loss |
4 (2%) |
2 (2%) |
|
Decreased activity, lethargy |
4 (2%) |
1 (1%) |
|
Vomiting |
4 (2%) |
1 (1%) |
|
Cystitis, hematuria |
3 (2%) |
0 (0%) |
|
Hematochezia, diarrhea |
3 (2%) |
1 (1%) |
|
Hair loss, excoriation, bruising |
2 (1%) |
0 (0%) |
|
Respiratory, cardiac arrest |
1 (1%) |
0 (0%) |
|
Incoordination, weakness |
1 (1%) |
1 (1%) |
|
Death |
0 (0%) |
1 (1%) |
*Cats may have experienced more than one of these signs during the study.
One cat was reported to have vomiting, diarrhea, incoordination, tachypnea, weakness, and tachycardia approximately 24 h following the second administration of ONSIOR. On physical examination, the cat was described as very weak and uncoordinated, which was attributed to hypoxia. With treatment, the cat gradually recovered the same day. Blood work was within normal limits.
While pain was not reported as an adverse reaction, the most common reasons associated with “rescue due to pain” as identified by investigators were tenderness of surgical sites, aggressive/guarding behaviour, vocalizing, and agitated, purposeful avoidance of painful stimulus, hunched position, trembling/shaking, little or no social response, and tachycardia/tachypnea. (See EFFICACY STUDIES)
In four field studies, cats of various breeds, 10 months to 19 years old weighing 2.6 to 10.7 kg with chronic musculoskeletal disorders or osteoarthritis were treated with ONSIOR or placebo for 3 to 12 weeks. The most commonly reported clinical signs (vomiting/emesis, anorexia, diarrhea and lethargy) were seen with similar frequency in cats treated with either ONSIOR or placebo.
Adverse Events in the Chronic Musculoskeletal Disorder or Osteoarthritis Field Studies
|
Adverse Event* |
ONSIOR 6 mg Tablets (n=267 cats) |
Placebo Control (n=271 cats) |
|
Vomiting/emesis |
55 (20.6%) |
45 (16.6%) |
|
Anorexia |
16 (6.0%) |
10 (3.7%) |
|
Diarrhea |
10 (3.7%) |
14 (5.2%) |
|
Lethargy |
8 (3.0%) |
16 (5.9%) |
|
Abnormal test finding |
6 (2.2%) |
7 (2.6%) |
|
Urine abnormality |
6 (2.2%) |
3 (1.1%) |
*Cats may have experienced more than one of these signs during the study.
There were no statistically significant and clinically relevant differences between the robenacoxib and placebo groups in the frequency of adverse events or in any clinical pathological variable, including hepatic or renal biochemistry.
It was concluded that ONSIOR was well-tolerated by most cats for periods of 3 to 12 weeks.
EFFICACY STUDIES:
1. For the relief of acute pain and inflammation associated with cat bites and scratches with and without abscesses and musculoskeletal injuries such as sprains and strains in cats.
Efficacy was demonstrated using ONSIOR in a blinded, positive controlled, multi-site field study conducted in France and the United Kingdom involving client-owned cats. In this study cats with signs of acute musculoskeletal pain and inflammation caused by injury or a bite with or without an abscess were randomly assigned to receive 5 or 6 days of treatment with ONSIOR tablets or the positive control article. There were 56 cats in the ONSIOR tablets group and 48 cats in the positive control group. The primary endpoint was a global investigator score, a composite score of pain on palpation, inflammation and mobility. Non-inferiority was demonstrated for the primary endpoint. 63% of cats had concomitant treatments of antibiotics, lancing and draining of abscesses. The results of the study demonstrated the efficacy of ONSIOR tablets was non-inferior to the positive control treatment.
2. For the control of postoperative pain and inflammation associated with onychectomy, ovariohysterectomy, and castration in cats.
Efficacy was demonstrated using ONSIOR in a masked, controlled, multi-site field study. In this study, 249 cats presenting for ovariohysterectomy or castration in conjunction with an onychectomy (forelimbs only) were randomly administered ONSIOR in the active treatment group, or a placebo in the control treatment group. There were 167 cats in the ONSIOR group and 82 cats in the control group. Cats treated with ONSIOR, butorphanol and a metacarpal 4 point ring block using bupivacaine were compared to cats treated with a placebo tablet, butorphanol and a metacarpal 4 point ring block using bupivacaine on the need to provide rescue medication to control postoperative pain. The cats were assessed at extubation, and at 30 minutes, 1h, 3h, 5h, 8h, 24h, 28h, 32h, 48h and 52 h post extubation. The drug was administered approximately 30 minutes prior to surgery along with pre-anesthetic medications and continued once daily for two additional treatments. Effectiveness was evaluated in 244 cats and field safety was evaluated in 249 cats. A statistically significant difference in the proportion of treatment successes in the ONSIOR treatment group (137/164 or 83.5%) compared to the control treatment group (43/80 or 53.8%) was observed. Twenty-seven out of 164 ONSIOR cases (16.5%) and 37 out of 80 control treatment cases (46.2%) were treatment failures. The most common reasons checked by investigators for rescue were tenderness of surgical sites, aggressive/guarding behaviour, vocalizing, and agitated, purposeful avoidance of painful stimulus, hunched position, trembling/shaking, little or no social response, and tachycardia/tachypnea. Statistically significant differences for pain elicited on palpation at the spay or castration incision site, behaviour following social interaction and posture score at various post-surgical time points were also observed. The results of the field study demonstrate that ONSIOR, when administered for a maximum of three days, is effective and well-tolerated as an adjunctive medication in the control of postoperative pain associated with onychectomy, ovariohysterectomy, and castration in cats.
3. For the control of pain and inflammation associated with chronic musculoskeletal disorders, such as osteoarthritis, in cats.
Efficacy was demonstrated using ONSIOR in two related masked, controlled, multi-site field studies. A total of 140 cats with chronic musculoskeletal disorder were enrolled at eight clinical sites in the US. Included cats had to have a history of impaired mobility plus pain and radiographic signs of degenerative joint disease in at least two joints. The cats were aged 2 to 18 years with body weight 3.2 to 10.7 kg. The cats were administered ONSIOR as the active treatment (R) or placebo (P) via random allocation to one of three sequence groups: PPP, n=47; PRR, n=47; and PRP, n=46. The three phases of the study had durations respectively of 2, 3 and 3 weeks. From the 140 cats enrolled in both studies, the final data sets were safety, n=138 and efficacy n=136.
Compared to placebo, ONSIOR was effective in significantly increasing activity (assessed objectively using activity monitors) and improving subjective scores made by owners of behaviour, quality of life, temperament and happiness. The size of the treatment effect of ONSIOR compared to placebo was an approximately 5% increase in activity over 24 hours (day-night) and 10% at night-time. For the subjective client-specific outcome measures (CSOM) over the 6 week treatment period, mean values were 6.5 (placebo) and 7.6 (ONSIOR); the difference of 1.1 represents an improvement of approximately 17% and was significant (P=0.0195). The CSOM was the sum of three activities which were scored as 4 (no problem), 3 (mild difficulty), 2 (moderate difficulty), 1 (severe difficulty) or 0 (impossible); the range was therefore 0-12 with lower scores indicating greater impairment.
The results of the field studies demonstrate that ONSIOR, when administered for 3 or 6 weeks, is effective and well-tolerated in the control of pain and inflammation associated with chronic musculoskeletal disorders in cats.
In two earlier field studies no significant differences were detected between placebo and ONSIOR; reasons for the failure include inappropriate case selection, lack of specificity and sensitivity of the simple subjective scoring schemes used, no baseline period and a high care-giver placebo effect.
STORAGE CONDITION:
ONSIOR tablets should be stored between 5°C and 25°C.
PRESENTATION:
ONSIOR tablets are available in a 6 mg tablet strength for oral administration.
Date: April 2023
Elanco Canada Limited, 1919 Minnesota Court, Suite 401, Mississauga, Ontario L5N 0C9
Onsior, Elanco and the diagonal bar logo are trademarks of Elanco or its affiliates.
© 2023 Elanco or its affiliates.
04Apr2023
CPN: 1231105.8
Copyright © 2023 Animalytix LLC. Updated: 2023-11-27

(robenacoxib)
6 mg tablets for cats
Non-Steroidal Anti-Inflammatory
For Veterinary Use Only
For Use in Cats Only
DIN 02375834
Description
ONSIOR contains robenacoxib which is a non-steroidal anti-inflammatory drug (NSAID) belonging to the coxib class. The flavoured tablets are round, beige to brown in colour and are not scored.
Onsior (tablets for cats) (6 mg) Indications
ONSIOR tablets are indicated:
1. for the relief of acute pain and inflammation associated with cat bites and scratches with and without abscesses and musculoskeletal injuries such as sprains and strains in cats.
2. as an adjunctive medication, in the control of postoperative pain and inflammation associated with onychectomy, ovariohysterectomy, and castration in cats.
3. for the control of pain and inflammation associated with chronic musculoskeletal disorders, such as osteoarthritis, in cats.
Dosage and Administration
For oral use in cats ≥ 2.5 kg AND ≥ 4 months of age.
Carefully consider the potential benefits and risks of ONSIOR and other treatment options before deciding to use ONSIOR (robenacoxib). Use the lowest effective dose for the shortest duration consistent with individual response.
Give orally without food or with a small quantity of food (see PHARMACOLOGY).
The recommended daily dose of ONSIOR is 1 mg/kg body weight with a range 1 to 2.4 mg/kg (see Dosing Chart below).
For relief of acute pain and inflammation associated with cat bites and scratches with and without abscesses and musculoskeletal injuries, administer once per day for a maximum of 6 days according to the dosing chart below.
As an adjunctive medication, in the control of postoperative pain and inflammation associated with onychectomy, ovariohysterectomy and castration, administer once per day for a maximum of 3 days according to the dosing chart below. The first dose should be administered approximately 30 minutes (without food) before the start of surgery, around the time of induction of general anesthesia.
After surgery, once daily treatment may be continued for up to two days with either ONSIOR tablets or ONSIOR injection at their respective label recommended dose. If subsequent doses are given by subcutaneous injection, different sites for each injection should be used. If necessary, additional analgesic treatment with opioids is recommended.
For the control of pain and inflammation associated with chronic musculoskeletal disorders, such as osteoarthritis, in cats, administer once daily according to the table below. Duration of treatment should be decided on an individual basis. Response to treatment should be monitored at regular intervals by the responsible veterinarian. In clinical field studies, robenacoxib was well-tolerated by most cats for periods up to 12 weeks. A clinical response is normally seen within 3 to 6 weeks. Treatment should be discontinued after 6 weeks if no clinical improvement is apparent.
Note: In cats, the dose of ONSIOR tablets and ONSIOR injectable are different.
Dosing Chart:
|
Body Weight |
ONSIOR (robenacoxib) 6 mg Tablets / Frequency |
|
2.5 to less than 6 kg |
1 whole tablet once daily |
|
6 to less than 12 kg |
2 whole tablets once daily |
Tablets are not scored and should not be broken.
Do not use in cats weighing less than 2.5 kg as these animals cannot be accurately dosed.
As with other NSAIDs, do not exceed the recommended dosage (see CONTRAINDICATIONS and CAUTIONS sections). When prescribing NSAIDs, exceeding the label recommended dose could lead to complications, including acute renal failure and in some cases associated death has been reported.
Owners should be advised when their pet has received a robenacoxib injection prior to initiating the use of ONSIOR tablets, and be informed of the potential for adverse reactions and clinical signs associated with possible NSAID intolerance. Always provide the Client Information Sheet with prescription (detach from Package Insert).
Contraindications
As with all non-steroidal anti-inflammatory drugs (NSAIDs), administration of this drug is contraindicated in the following circumstances:
● cats with gastrointestinal ulcers;
● cats that are dehydrated, hypovolemic, hypoproteinemic or hypotensive;
● cats with impaired cardiac, renal or hepatic function, or coagulation abnormalities;
● a known hypersensitivity to robenacoxib or its excipients;
● concurrent use of other NSAIDs or corticosteroids;
● in breeding, pregnant or lactating cats because the safety of robenacoxib has not been established in these animals.
CAUTIONS:
The safety of ONSIOR tablets has not been established in cats weighing less than 2.5 kg or under 4 months of age.
Prolonged treatment with robenacoxib for the relief of chronic pain and inflammation should only be considered after a risk/benefit assessment by the responsible veterinarian.
All cats should undergo a thorough history and physical exam before the initiation of NSAID therapy. Appropriate laboratory tests should be conducted to establish hematological and biochemical baseline data before the administration of any NSAID. ONSIOR should be used with caution in cats with a known hypersensitivity to other NSAIDs.
As a class, cyclo-oxygenase inhibitory NSAIDs may be associated with gastrointestinal, renal, and hepatic toxicity. Sensitivity to drug-associated adverse events varies with the individual patient. Cats that have experienced adverse reactions from one NSAID may experience adverse reactions from another NSAID. Patients at greatest risk for adverse events are those that are dehydrated, on concomitant diuretic therapy, or those with existing renal, cardiovascular, and/or hepatic dysfunction. Anesthetic drugs may affect renal perfusion; approach concomitant use of anesthetics and NSAIDs cautiously. Appropriate monitoring procedures (including ECG, blood pressure, and temperature regulation) should be employed during all surgical procedures. The use of parenteral fluids during surgery is recommended to decrease potential renal complications when using NSAIDs perioperatively.
If decrease in appetite, vomiting, diarrhea, change in drinking or urination, change in behaviour, such as depression or restlessness, or other suspected adverse reactions occur, stop administration of ONSIOR immediately and seek the advice of a veterinarian (see ADVERSE REACTIONS).
Pre-treatment with other anti-inflammatory drugs may result in additional or increased adverse effects and accordingly a treatment-free period with such substances should be observed. The treatment-free period should take into account the pharmacokinetic properties of the products used previously.
Concomitant treatment with drugs which affect renal flow, e.g. anesthetics, should be subject to clinical monitoring. Concurrent administration of potentially nephrotoxic medicines should be avoided as there might be an increase of renal toxicity. The use of concomitantly protein-bound drugs with ONSIOR Tablets for Cats has not been studied in cats. Drug compatibility should be monitored closely in patients requiring additional therapy.
ONSIOR was administered concurrently with pre-anesthetic (opioid) and a metacarpal four point ring block using a longer acting local anesthetic medication, during the field study assessing the effect of ONSIOR on postoperative pain control. Level of analgesia should be monitored postoperatively to assess if additional pain control is needed. If necessary, additional analgesic treatment with opioids is recommended. Other NSAIDs and corticosteroids should not be used.
The use of ONSIOR in cats with cardiac disease has not been studied. ONSIOR has been shown to prolong the QT interval in repeat dose laboratory studies. In anaesthetised healthy cats, a single intravenous administration of 2 or 4 mg/kg ONSIOR had no effect on the ECG including the QT interval. The associated risk of developing a ventricular arrhythmia is unknown. The use of robenacoxib with other drugs shown to prolong the QT interval is not recommended. Commonly used drugs that prolong QT interval include antihistamines and prokinetic drugs. Appropriate monitoring procedures including ECG should be employed during all surgical procedures.
Studies to determine the activity of robenacoxib when administered concomitantly with other protein-bound or similarly metabolized drugs have not been conducted. Commonly used protein-bound drugs include cardiac, anticonvulsant, and behavioural medications. The influence of concomitant drugs that may inhibit metabolism of ONSIOR has not been evaluated. Drug compatibility should be monitored in patients requiring adjunctive therapy.
A gamma lactam (γ-lactam) metabolite of robenacoxib is formed in the cat. In addition, this γ-lactam is a degradation product that increases over the shelf life of the tablets and can be decreased by refrigeration. Neurological signs have been associated with the use of β lactam drugs; it is unknown if this γ-lactam may cause similar neurological signs.
Warnings
KEEP OUT OF REACH OF CHILDREN. In case of accidental ingestion, seek medical advice immediately and show the package insert or the label to the physician.
Adverse Reactions
Although all adverse events are not reported, the following information is based on voluntary post-approval drug experience reporting. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using this data.
The post-marketing adverse event reports for ONSIOR 6 mg tablets for cats have shown the following signs to be reported very rarely (less than 1 animal in 10,000 animals, including isolated reports): anorexia, lethargy, vomiting, and diarrhea, elevated creatinine, elevated BUN, and renal insufficiency.
For technical support or to report a suspected adverse drug reaction, contact Elanco Canada Limited at 1-800-265-5475.
Information for Cat Owners:
ONSIOR is the product your veterinarian has chosen to treat your cat’s pain and inflammation. It belongs to a class of drugs called “non-steroidal anti-inflammatory” or NSAIDs. These drugs must be used according to your veterinarian’s directions. Occasionally, NSAIDs can cause side effects. If your cat stops eating, is depressed, vomits or has diarrhea stop the drug immediately and contact your veterinarian. In most cases the side effects will disappear when the drug is stopped but in rare cases it may be serious. For this reason, it is important to consult with your veterinarian. If you notice any serious side effects or others not mentioned in the insert, please inform your veterinarian.
Pharmacology
Mode of Action
Pharmacodynamics
Robenacoxib is an NSAID of the coxib class. It is a highly potent and selective inhibitor of the cyclooxygenase 2 enzyme (COX-2). The cyclooxygenase enzyme (COX) is present in two forms. COX-1 is the “constitutive” form of the enzyme and has protective functions including in the gastrointestinal tract and kidney. COX-2 is the “inducible” form of the enzyme and is responsible for the production of mediators including PGE2 which induce pain, inflammation or fever.
Pharmacokinetics
Absorption: In a feline pharmacokinetic study, estimates for PK parameters in fasted cats included Cmax 1158.9 ng/mL, Tmax 0.5 h, AUC (0-∞) 1337 ng.h/mL. Co-administration with the normal diet (1/3 of daily ration) produced no change in Cmax, Tmax or AUC (0-∞). Co-administration with the entire daily ration of food produced no change in Tmax but a 20% reduction in AUC (0-∞) and a 40.3% reduction in Cmax of robenacoxib.
The systemic bioavailability of non-flavoured robenacoxib tablets was 49% without food.
Distribution: Robenacoxib has a relatively small volume of distribution (Vss 190 mL/kg) and is highly bound to plasma proteins (>98%).
Biotransformation: Robenacoxib is extensively metabolized by the liver in cats. The systemic exposure of the γ-lactam metabolite is about 25% of robenacoxib exposure following oral administration to fed cats. Further, the systemic exposure to lactam appears to be two-fold greater in fed cats than in fasted cats (see CAUTIONS). Apart from the one γ-lactam metabolite, the identity and activity of the other metabolites is not known in cats.
Elimination: Robenacoxib is rapidly cleared from blood (CL 0.44 L/kg/h) with a mean elimination half-life 1.1 h after intravenous administration. After oral administration of tablets, the mean terminal half-life from blood was 1.7 h, and elimination is predominantly through the biliary route (fecal and urinary excretion are 60 and 16.5% respectively).
The pharmacokinetics of robenacoxib do not differ between male and female cats.
SAFETY STUDIES:
21-Day Target Animal Safety Study:
In a 21-day laboratory tolerance study, 8 month-old healthy, DSH cats (4/sex/group) were administered robenacoxib at a dose of 0 mg/kg (control group) or 24 mg/kg/day (10X the maximum exposure based on the single, 6 mg tablet size). All cats survived to study termination. Vomiting and decreased activity was noted in some of the treated cats. Two cats in the 10X group exhibited abnormal rear limb neurologic function. One of these cats also exhibited a head tilt and nystagmus at the end of the study. Mean food consumption was less in the 10X group. The mean kidney weights were lower in the 10X group compared to the control group; and the mean thymus weights were also lower in the 10X group compared to the controls. Two cats in the 10X group had chronic interstitial nephritis on histopathology; this finding was correlated with a renal mass observed on gross necropsy in one cat. One 10X cat had a focal cecal/large intestinal erosion. One 10X cat and one control cat had periportal, multifocal necrosis in one lobe of the liver. There were four 10X cats and 2 control cats with renal tubular degeneration. Under the conditions of this study, robenacoxib was well tolerated when administered at 24 mg/kg/day for 21 days, except for 2 cats in the 10X group with neurologic signs.
42-Day Target Animal Safety Study:
In a 42-day study, 8 month-old, healthy cats were administered robenacoxib at 0, 2, 6 or 10 mg/kg/twice daily. Small thymuses were noted in all robenacoxib-treated groups with corresponding organ weight decreases and/or atrophic changes on histopathology. There was a decrease in the kidney weights in the 10 mg/kg/twice daily-group compared to the controls. Vomiting was the most common adverse reaction noted in the treated cats. An adequate safety margin was demonstrated for ONSIOR when administered under the conditions of this 42-day study.
6 Month Target Animal Safety Study:
In a 6 month study, 8 month old, healthy cats (4/sex/group) were administered robenacoxib at 0, 1X (2.4 mg/kg), 3X (7.2 mg/kg) or 5X (12 mg/kg) once daily. One 5X cat had clonic seizures on Day 115 and was ataxic on Day 175. One 5X cat had skin cold to the touch on Day 106. One cat in the 1X group experienced urethral obstruction/FLUTD. Vomiting, decreased activity, injected sclera and soft stools were the most common adverse reactions observed in the treated groups. Soft stools and injected sclera were also observed in the control group.
The mean body weights in the 1X and 3X groups were lower than the controls from Day 21 to Day 182; and lower in the 5X group from Day 28 to Day 182. There was a clear dose-related and possibly time-related increase in the QTc interval at Day 41 and Day 175, particularly in the 3X and 5X groups, no other ECG abnormalities were noted.
There was no obvious accumulation in Cmax or AUC between Days 1, 31 and 171, and there was no apparent difference in parameters between males and females. The following parameters were calculated for the 1X dosage: Tmax was 0.5 h (median), the dose-normalized mean Cmax was 668 ng/mL and the dose-normalized mean area under the curve (AUC(0-inf)) was 902 h*ng/mL. Similarly, the following parameters were calculated for the 3X dosage: Tmax was 0.5 h (median), the dose normalized mean Cmax was 1019 ng/mL and the dose-normalized mean area under the curve (AUC(0-inf)) was 1394 h*ng/mL. For the 5X dosage the following parameters were calculated: Tmax was 1.0 h (median), the dose-normalized mean Cmax was 1198 ng/mL and the dose-normalized mean area under the curve (AUC(0-inf)) was 1884 h*ng/mL. A post hoc analysis of PK parameters revealed that dose normalized Cmax and AUC were greater than dose proportional.
The mean kidney weights were lower in all robenacoxib-treated groups. One 5X cat with decreased kidney weight and size also had transient increases in BUN and creatinine. There were transient increases in AST, amylase, and ALT in the 3X and 5X cats from Day 30 to Day 183. There was an increased severity of the tubular degeneration/regeneration in the kidneys of two 1X and two 5X cats with inflammation, papillary necrosis and papillary mineralization. An increased incidence of minimal to mild Kupffer cell pigmentation was observed in the livers of all robenacoxib-treated cats; however, no hepatocellular damage was noted on histopathology. One 5X cat had a focal, minimal ulcer of the gastric fundus (peptic and parietal cells).
This 6 month safety study supports the safe use of ONSIOR for the relief of acute pain and inflammation associated with cat bites and scratches with and without abscesses and musculoskeletal injuries such as sprains and strains in cats for a maximum of 6 days.
37-Day Interchangeable Use Study:
ONSIOR was administered orally (6 mg tablets) and subcutaneously (20 mg/mL solution) to 4-month old healthy cats at 0, 1, 2, and 3 times the labeled doses (1X = 2.4 mg/kg/day orally based on the inherent tablet dose band or 2 mg/kg/day subcutaneously). Interchangeable use was evaluated by alternating three 7-day oral tablet/3-day subcutaneous injection cycles followed by one final 7-day oral tablet dosing cycle. Findings included: elevated creatine kinase levels on Days 13 and 37, soft stools, histologic observation of a minimal oral (tongue) ulceration in a 1X cat, injection site edema for up to 120 h prior to resolution, and a prolonged QT interval in treated cats as compared to the controls on Day 36. Histologically, the injection site had minimal or mild, subacute/chronic inflammation. Inflammation at the injection site was observed in both treated and control animals with a greater frequency in the higher dose groups than in the control and 1X groups. One male and one female in the 1x group had focal tubular degeneration/regeneration of the renal cortex. Focal tubular degeneration/regeneration represents non-specific renal findings which can occur as a result of many causes. Dose-normalized AUC and concentration levels were higher following the oral route than the subcutaneous route. There was no significant accumulation following once daily administration. One 2X-treated cat had a 7-fold increase in buccal mucosal bleeding time (BMBT) during the treatment period compared to the pre-treatment value.
Preliminary 37-Day Interchangeable Use Study:
ONSIOR was administered orally (6 mg tablets) and subcutaneously (20 mg/mL solution) to 4-month old cats at 0, 1, and 5 times the labeled doses (1X = 2.4 mg/kg/day orally based on the inherent tablet dose band or 2 mg/kg/day subcutaneously). Interchangeable use was evaluated by alternating three 7-day oral tablet/3-day subcutaneous injection cycles followed by one final 7-day oral tablet dosing cycle. Clinical findings included: scabs and sores at the injection sites of one 1X female and two 5X females, and injection site edema noted more frequently in treated cats. Injection site changes were characterized as minimal to moderate granulomatous inflammation, minimal to moderate fibroplasia/fibrosis, and minimal myofiber regeneration of the panniculus carnosus. In one 1X female, moderate necrosis of a blood vessel was noted within the granulomatous inflammation. Minimal myofiber regeneration was observed in the underlying skeletal muscle in three out of four 5X males.
A red depressed area on the upper lip of one 5X cat correlated histologically with a minimal ulcer. Creatinine was significantly increased in 5X cats compared to the controls. Urine specific gravities remained within normal limits for all 5X cats, and blood urea nitrogen (BUN) values remained within normal limits for all study animals. Histologically, renal changes included bilateral or unilateral minimal to moderate vacuolation and bilateral or unilateral minimal to mild degeneration of proximal tubules were observed in three 5X males. Two 5X males had mineralized foci in the epithelium covering the papilla.
One 5X female had a brief episode of ataxia and lethargy on Day 16. This cat was subsequently noted to be dehydrated and constipated, requiring veterinary intervention with subcutaneous fluid therapy and nutritional supplementation. This cat had the greatest QT increase on ECG evaluation.
Clinical Safety
In the pivotal clinical trial to evaluate acute pain and inflammation associated with musculoskeletal disorders, where 56 cats were treated with ONSIOR, diarrhea, emesis, lethargy, hyperactivity and polydipsia were reported at the following frequencies:
Adverse Reactions in the Acute Musculoskeletal Disorder Field Study
|
Adverse Reactions |
ONSIOR 6 mg Tablets |
Positive Control |
|
Diarrhea |
4 (7%) |
1 (2%) |
|
Emesis |
3 (5%) |
0 (0%) |
|
Lethargy |
3 (5%) |
1 (2%) |
|
Hyperactivity |
2 (4%) |
0 (0%) |
|
Polydipsia |
1 (2%) |
0 (0%) |
In a placebo controlled postoperative pain field study associated with ovariohysterectomy, castration and onychectomy, 249 male and female cats of various breeds, 6 months to 13 years old weighing 2.5 to 7.5 kg, the most commonly reported adverse reactions were surgical site bleeding, infected surgery sites, lethargy, vomiting and inappetence.
Adverse Reactions in the Postoperative Pain Field Study
|
Clinical Sign* |
ONSIOR 6 mg Tablets (n=167 cats) |
Positive Control (n=82 cats) |
|
Incision site bleeding |
7 (4%) |
1 (1%) |
|
Incision site infection |
6 (4%) |
2 (2%) |
|
Inappetence, weight loss |
4 (2%) |
2 (2%) |
|
Decreased activity, lethargy |
4 (2%) |
1 (1%) |
|
Vomiting |
4 (2%) |
1 (1%) |
|
Cystitis, hematuria |
3 (2%) |
0 (0%) |
|
Hematochezia, diarrhea |
3 (2%) |
1 (1%) |
|
Hair loss, excoriation, bruising |
2 (1%) |
0 (0%) |
|
Respiratory, cardiac arrest |
1 (1%) |
0 (0%) |
|
Incoordination, weakness |
1 (1%) |
1 (1%) |
|
Death |
0 (0%) |
1 (1%) |
*Cats may have experienced more than one of these signs during the study.
One cat was reported to have vomiting, diarrhea, incoordination, tachypnea, weakness, and tachycardia approximately 24 h following the second administration of ONSIOR. On physical examination, the cat was described as very weak and uncoordinated, which was attributed to hypoxia. With treatment, the cat gradually recovered the same day. Blood work was within normal limits.
While pain was not reported as an adverse reaction, the most common reasons associated with “rescue due to pain” as identified by investigators were tenderness of surgical sites, aggressive/guarding behaviour, vocalizing, and agitated, purposeful avoidance of painful stimulus, hunched position, trembling/shaking, little or no social response, and tachycardia/tachypnea. (See EFFICACY STUDIES)
In four field studies, cats of various breeds, 10 months to 19 years old weighing 2.6 to 10.7 kg with chronic musculoskeletal disorders or osteoarthritis were treated with ONSIOR or placebo for 3 to 12 weeks. The most commonly reported clinical signs (vomiting/emesis, anorexia, diarrhea and lethargy) were seen with similar frequency in cats treated with either ONSIOR or placebo.
Adverse Events in the Chronic Musculoskeletal Disorder or Osteoarthritis Field Studies
|
Adverse Event* |
ONSIOR 6 mg Tablets (n=267 cats) |
Placebo Control (n=271 cats) |
|
Vomiting/emesis |
55 (20.6%) |
45 (16.6%) |
|
Anorexia |
16 (6.0%) |
10 (3.7%) |
|
Diarrhea |
10 (3.7%) |
14 (5.2%) |
|
Lethargy |
8 (3.0%) |
16 (5.9%) |
|
Abnormal test finding |
6 (2.2%) |
7 (2.6%) |
|
Urine abnormality |
6 (2.2%) |
3 (1.1%) |
*Cats may have experienced more than one of these signs during the study.
There were no statistically significant and clinically relevant differences between the robenacoxib and placebo groups in the frequency of adverse events or in any clinical pathological variable, including hepatic or renal biochemistry.
It was concluded that ONSIOR was well-tolerated by most cats for periods of 3 to 12 weeks.
EFFICACY STUDIES:
1. For the relief of acute pain and inflammation associated with cat bites and scratches with and without abscesses and musculoskeletal injuries such as sprains and strains in cats.
Efficacy was demonstrated using ONSIOR in a blinded, positive controlled, multi-site field study conducted in France and the United Kingdom involving client-owned cats. In this study cats with signs of acute musculoskeletal pain and inflammation caused by injury or a bite with or without an abscess were randomly assigned to receive 5 or 6 days of treatment with ONSIOR tablets or the positive control article. There were 56 cats in the ONSIOR tablets group and 48 cats in the positive control group. The primary endpoint was a global investigator score, a composite score of pain on palpation, inflammation and mobility. Non-inferiority was demonstrated for the primary endpoint. 63% of cats had concomitant treatments of antibiotics, lancing and draining of abscesses. The results of the study demonstrated the efficacy of ONSIOR tablets was non-inferior to the positive control treatment.
2. For the control of postoperative pain and inflammation associated with onychectomy, ovariohysterectomy, and castration in cats.
Efficacy was demonstrated using ONSIOR in a masked, controlled, multi-site field study. In this study, 249 cats presenting for ovariohysterectomy or castration in conjunction with an onychectomy (forelimbs only) were randomly administered ONSIOR in the active treatment group, or a placebo in the control treatment group. There were 167 cats in the ONSIOR group and 82 cats in the control group. Cats treated with ONSIOR, butorphanol and a metacarpal 4 point ring block using bupivacaine were compared to cats treated with a placebo tablet, butorphanol and a metacarpal 4 point ring block using bupivacaine on the need to provide rescue medication to control postoperative pain. The cats were assessed at extubation, and at 30 minutes, 1h, 3h, 5h, 8h, 24h, 28h, 32h, 48h and 52 h post extubation. The drug was administered approximately 30 minutes prior to surgery along with pre-anesthetic medications and continued once daily for two additional treatments. Effectiveness was evaluated in 244 cats and field safety was evaluated in 249 cats. A statistically significant difference in the proportion of treatment successes in the ONSIOR treatment group (137/164 or 83.5%) compared to the control treatment group (43/80 or 53.8%) was observed. Twenty-seven out of 164 ONSIOR cases (16.5%) and 37 out of 80 control treatment cases (46.2%) were treatment failures. The most common reasons checked by investigators for rescue were tenderness of surgical sites, aggressive/guarding behaviour, vocalizing, and agitated, purposeful avoidance of painful stimulus, hunched position, trembling/shaking, little or no social response, and tachycardia/tachypnea. Statistically significant differences for pain elicited on palpation at the spay or castration incision site, behaviour following social interaction and posture score at various post-surgical time points were also observed. The results of the field study demonstrate that ONSIOR, when administered for a maximum of three days, is effective and well-tolerated as an adjunctive medication in the control of postoperative pain associated with onychectomy, ovariohysterectomy, and castration in cats.
3. For the control of pain and inflammation associated with chronic musculoskeletal disorders, such as osteoarthritis, in cats.
Efficacy was demonstrated using ONSIOR in two related masked, controlled, multi-site field studies. A total of 140 cats with chronic musculoskeletal disorder were enrolled at eight clinical sites in the US. Included cats had to have a history of impaired mobility plus pain and radiographic signs of degenerative joint disease in at least two joints. The cats were aged 2 to 18 years with body weight 3.2 to 10.7 kg. The cats were administered ONSIOR as the active treatment (R) or placebo (P) via random allocation to one of three sequence groups: PPP, n=47; PRR, n=47; and PRP, n=46. The three phases of the study had durations respectively of 2, 3 and 3 weeks. From the 140 cats enrolled in both studies, the final data sets were safety, n=138 and efficacy n=136.
Compared to placebo, ONSIOR was effective in significantly increasing activity (assessed objectively using activity monitors) and improving subjective scores made by owners of behaviour, quality of life, temperament and happiness. The size of the treatment effect of ONSIOR compared to placebo was an approximately 5% increase in activity over 24 hours (day-night) and 10% at night-time. For the subjective client-specific outcome measures (CSOM) over the 6 week treatment period, mean values were 6.5 (placebo) and 7.6 (ONSIOR); the difference of 1.1 represents an improvement of approximately 17% and was significant (P=0.0195). The CSOM was the sum of three activities which were scored as 4 (no problem), 3 (mild difficulty), 2 (moderate difficulty), 1 (severe difficulty) or 0 (impossible); the range was therefore 0-12 with lower scores indicating greater impairment.
The results of the field studies demonstrate that ONSIOR, when administered for 3 or 6 weeks, is effective and well-tolerated in the control of pain and inflammation associated with chronic musculoskeletal disorders in cats.
In two earlier field studies no significant differences were detected between placebo and ONSIOR; reasons for the failure include inappropriate case selection, lack of specificity and sensitivity of the simple subjective scoring schemes used, no baseline period and a high care-giver placebo effect.
STORAGE CONDITION:
ONSIOR tablets should be stored between 5°C and 25°C.
PRESENTATION:
ONSIOR tablets are available in a 6 mg tablet strength for oral administration.
Date: April 2023
Elanco Canada Limited, 1919 Minnesota Court, Suite 401, Mississauga, Ontario L5N 0C9
Onsior, Elanco and the diagonal bar logo are trademarks of Elanco or its affiliates.
© 2023 Elanco or its affiliates.
04Apr2023
CPN: 1231105.8
Copyright © 2023 Animalytix LLC. Updated: 2023-11-27
Онсиор ─ это препарат, который используется в ветеринарной практике для устранения воспаления и болевого синдрома у кошек. Он всасывается в желудочно-кишечном тракте, после чего поступает в системный кровоток.
Содержание
- 1 Состав
- 2 Формы выпуска
- 3 Показания и противопоказания
- 4 Побочные эффекты
- 5 Инструкция по применению
- 6 Совместимость с другими препаратами
- 7 Как хранить
- 8 О мерах предосторожности
- 9 Цена
- 10 Отзывы владельцев
- 11 Отзывы ветеринаров
Состав
Медикамент относится к нестероидным противовоспалительным средствам из группы коксибов. Основное действующее вещество— Робенакоксиб. Также, присутствуют вспомогательные компоненты: стеарат магния; дрожжи; повидон; кросповидон и другие.
Формы выпуска
Препарат выпускается в двух формах:
- Таблетки круглой формы, коричневого или бежевого цвета в блистерах по 6 штук.
- Раствор для инъекций.
Показания и противопоказания
Показания для применения Онсиора у кошек:
- вывихи и раны;
- воспаление в мягких тканях;
- болезни суставов (артрозы или артриты у кошек).
Также, препарат можно использовать в качестве премедикации перед выполнением операции (примерно за пол часа до введения наркотического вещества).
Онсиор категорически противопоказан к применению у беременных и лактирующих кошек. Другие противопоказания:
- хронические заболевания печени, почек и сердца;
- состояние сильного обезвоживания;
- возраст (котята младше 4-х месяцев);
- индивидуальная непереносимость или повышенная чувствительность к компонентам препарата;
- вес животного меньше двух с половиной килограммов.
Побочные эффекты
Если соблюдать рекомендованную ветеринаром дозировку, то побочных явлений обычно не наблюдается.
При передозировке медикамента могут возникать следующие неприятные последствия:
- рвота;
- проблемы со стулом;
- симптомы почечной недостаточности;
- симптомы печеночной недостаточности.
Обычно, подобная симптоматика проходит самостоятельно и не требует дополнительного применения лекарств. Если у кошки проявляется аллергия на Онсиор, то врач назначает препараты с антигистаминным эффектом.
Инструкция по применению
Лекарство лучше давать отдельно от кормления. Если такой возможности нет, то его добавляют в корм. Рекомендованная дозировка— 1 таблетка в день на вес от 2,5 до 6 кг, 2 таблетки— на вес от 6 до 12 кг.
При наличии острых болей, лекарственно средство применяется 1 или 2 раза с интервалом в сутки. При заболеваниях суставов допускается принимать Онсиор в течение 7 дней.
При проведении хирургического вмешательства медикамент вводят в дозе 1-2 мг на один килограмм массы тела кошки.
Совместимость с другими препаратами
Онсиор нельзя употреблять одновременно с некоторыми другими медикаментами. К ним относятся:
- ингибиторы АПФ;
- диуретики;
- нестероидные противовоспалительные средства.
Все вышеперечисленные лекарства могут усилить токсическое действие Онсиора.
Как хранить
Таблетки необходимо хранить в сухом и прохладном месте. Оно должно быть надежно защищено от ультрафиолетовых лучей. Рекомендуемый температурный диапазон— 0-25 градусов.
О мерах предосторожности
При применении лекарства нужно соблюдать определенную технику безопасности. При случайном контакте Онсиора с кожными покровами или слизистыми оболочками, следует промыть их водой. Пустую тару из под медикамента категорически запрещено использовать в бытовых целях.
Цена
Купить Онсиор можно в аптеках Москвы, СПб и других городов. Стоимость упаковки таблеток 6 мг 6 штук— 550-600 рублей, раствора для инъекций фл.20 мл—2200-2400 рублей.
Посмотреть актуальную цену препарата и купить его теперь можно прямо здесь с Яндекс Маркета:
Отзывы владельцев
Наталья Сергеева:
«Моя кошка выпала на даче из окна второго этажа. В результате, была травмирована задняя лапа, и она начала прихрамывать. Я отвезла ее в клинику к ветеринарному врачу для осмотра. Специалист диагностировал вывих и порекомендовал подавать ей препарат Онсиор в течение двух или трех дней, чтобы уменьшить болевые ощущения и снять воспаление. Я купила лекарство в аптеке, и давала кошке по одной таблетке в день трое суток подряд. Побочных эффектов от употребления данного медикамента не заметила».
Мария Нестеренко:
«Моей кошке уже много лет, недавно она начала страдать артрозом тазобедренных суставов. На очередном приеме в ветеринарной клинике, врач посоветовал давать кошке Онсиор при выраженных болях. С тех пор, иногда использую его, если животное начинает ощущать дискомфорт и сильно беспокоится. Но это лишь вспомогательная мера, которая помогает быстро устранить болевой синдром. Основное лечение у нас другое. Специалист предупредил, что медикамент нельзя принимать на постоянной основе из-за риска возникновения побочных эффектов».
Отзывы ветеринаров
Виктория Антипенко:
«Я давно использую препарат Онсиор в своей работе. Он хорошо подходит для устранения болей при вывихах и патологиях опорно-двигательного аппарата у кошек. Лекарство действует быстро и эффективно. Также, его можно применять перед проведением оперативного вмешательства в качестве премедикации. Но назначать медикамент нужно аккуратно, потому что есть ряд противопоказаний».
Владимир Пирогов:
«В своей ветеринарной практике часто назначаю кошкам Онсиор при хронических заболеваниях суставов или травмах. Он хорошо устраняет болевой синдром и снимает воспаление. Но следует отметить, что иногда у животных встречается непереносимость компонентов препарата. В таких случаях, я отменяю данный медикамент, заменяя его другими альтернативными средствами».
Читайте также:
- Противосудорожный препарат для кошек Габапентин
- Обезболивающие препараты для кошек
- Судороги у кошек: причины и что делать
Загрузка…
ОНСИОР – лекарственный препарат для кошек и собак для лечения воспалительных и болевых синдромов различного происхождения, в том числе являющихся последствиями хирургического вмешательства.
Перед применением рекомендуется проконсультироваться с ветеринарным врачом.
Показания:
- Воспалительных и болевых синдромах различного происхождения.
- Острые и хронические заболевания опорно-двигательного аппарата (артриты, артрозы, синовиты, вывихи).
- Воспалительные заболевания мягких тканей.
- Послеоперационный период.
Общие сведения:
Торговое наименование лекарственного препарата: Онсиор таблетки (Onsior tablets).
Международное непатентованное наименование: робенакоксиб.
Лекарственная форма: таблетки для орального применения.
Онсиор в качестве действующего вещества содержит робенакоксиб.
Выпускают препарат в двух модификациях:
- Онсиор таблетки для кошек с содержанием робенакоксиба — 6 мг/табл
- Онсиор таблетки для собак в 4 дозировках с содержанием робенакоксиба: 5 мг/табл., 10 мг/табл., 20 мг/табл. или 40 мг/табл.
В качестве вспомогательных веществ препарат Онсиор содержит: дрожжи, целлюлозу микрокристаллическую, ароматизатор идентичный говядине (в «Онсиор таблетки для собак»), целлюлозу порошкообразную (в «Онсиор таблетки для собак»), повидон (К-30), кросповидон, двуокись кремния коллоидный безводный и стеарат магния.
По внешнему виду препарат представляет собой круглые таблетки от бежевого до коричневого цвета.
Онсиор таблетки для кошек и Онсиор таблетки для собак 5 мг имеют оттиск NA на одной стороне и оттиском АК с другой стороны;
Онсиор таблетки для собак 10 мг — оттиск NA на одной стороне и оттиск BE с другой стороны, Онсиор таблетки для собак 20 мг — оттиск NA на одной стороне и оттиск CD с другой стороны и Онсиор™ таблетки для собак 40 мг — оттиск NA на одной стороне и оттиск ВСК с другой стороны.
Срок годности лекарственного препарата при соблюдении условий хранения в закрытой упаковке производителя — 4 года с даты производства.
Запрещается применение Онсиор таблетки по истечении срока годности.
Онсиор таблетки для кошек выпускают расфасованным по 6 таблеток в блистеры из алюминиевой фольги, помещенные по 1, 2, 5 или 10 штук в картонные пачки вместе с инструкцией по применению.
Онсиор таблетки для собак выпускают расфасованным по 7 таблеток в блистеры из алюминиевой фольги, помещенные по 1, 2, 4 или 10 штук в картонные пачки вместе с инструкцией по применению.
Хранят таблетки в закрытой упаковке производителя в защищенном от прямых солнечных лучей месте, отдельно от пищевых продуктов и кормов, при температуре от 0°С до 25°С в местах, недоступных для детей.
Неиспользованный лекарственный препарат утилизируют в соответствии с требованиями законодательства.
Условия отпуска: без рецепта ветеринарного врача.
Фармакологические свойства:
Онсиор таблетки относится к нестероидным противовоспалительным лекарственным препаратам.
Робенакоксиб, входящий в состав препарата, — нестероидное противовоспалительное средство группы коксибов, является селективным специфическим ингибитором ЦОГ-2 и обладает противовоспалительным и анальгезирующим действием.
ЦОГ представлена в двух формах: ЦОГ-1 — конститутивная форма энзима — выполняет защитные функции, в том числе в желудочно-кишечном тракте и почках; ЦОГ-2 — индуцируемая форма энзима — отвечает за выработку медиаторов, включающих ПГЕг, вызывающих боль, воспаление и жар.
При пероральном введении робенакоксиб быстро всасывается в кишечнике, поступая в системный кровоток и большинство органов и тканей; более 99 % робенакоксиба связывается с белками плазмы. Его максимальная концентрация в плазме отмечается через 0,5 часа, биодоступность составляет 49 % у кошек и 62 % у собак при применении таблеток с кормом и 84 % без корма. Метаболизируется робенакоксиб в печени, выделяется в основном с желчью (около 70 % у кошек и около 65 % у собак) и частично — с мочой; период полувыведения из организма собак составляет около 1,2 часа, кошек — 1,7 часа.
Онсиор по степени воздействия на организм относится к умеренно опасным веществам (3 класс опасности по ГОСТ 12.1.007).
Порядок применения:
Онсиор таблетки назначают собакам и кошкам в качестве противовоспалительного и болеутоляющего препарата при воспалительных и болевых синдромах различного происхождения, включая острые и хронические заболевания опорно-двигательного аппарата (артриты, артрозы, синовиты, вывихи), воспалительные заболевания мягких тканей, а также в качестве анальгезирующего и противовоспалительного препарата в послеоперационный период.
Запрещено применять Онсиор животным в состоянии дегидратации, гиповолемии и гипотензии, а также животным массой менее 2,5 кг. При крайней необходимости назначения лекарственного препарата таким животным лечение проводят под постоянным наблюдением ветеринарного врача.
Противопоказанием к применению является индивидуальная повышенная чувствительность животного к компонентам препарата.
Не следует применять препарат животным с выраженной сердечной, почечной и печеночной недостаточностью.
Онсиор таблетки применяют собакам и кошкам индивидуально один раз в день в дозах, указанных в таблице:
|
Вид и масса животного |
Доза действующего вещества (мг/таблетка) и количество таблеток на животное |
||||
|
6 мг |
5 мг |
10 мг |
20 мг |
40 мг |
|
|
Кошки |
|||||
|
от 2,5 до 6 кг |
1 таб. |
||||
|
от 6 до 12 кг |
2 таб. |
||||
|
Собаки |
|||||
|
от 2,5 до 5 кг |
1 таб. |
||||
|
от 5 до 10 кг |
1 таб. |
||||
|
от 10 до 20 кг |
1 таб |
||||
|
от 20 до 40 кг |
1 таб. |
||||
|
от 40 до 80 кг |
2 таб. |
Рекомендуется давать лекарственный препарат отдельно от кормления, но, при необходимости, препарат может быть дан вместе с кормом. Таблетки не следует дробить или разламывать.
Онсиор таблетки применяют кошкам при оперативных вмешательствах за 30 минут до введения в наркоз в дозе 1-2,4 мг на 1 кг массы животного; лечение может быть продолжено в течение периода длительностью до 2 дней после операции; при заболеваниях опорно-двигательного аппарата препарат применяют до 6 дней.
Онсиор таблетки для собак применяют за 30 минут до или после кормления в дозе 1-2 мг на 1 кг массы животного. Курс лечения определяет лечащий ветеринарный врач. Как правило, продолжительность лечения составляет 7 дней. Если по истечении 10 дней клиническое улучшение не наступает, применение препарата прекращают.
При передозировке лекарственного препарата у животных с выраженной чувствительностью к компонентам препарата могут наблюдаться расстройство желудочно-кишечного тракта, печеночная или почечная недостаточность. Специфические средства детоксикации отсутствуют, применяют общие меры, направленные на выведение лекарственного препарата из организма.
Особенностей действия лекарственного препарата при его первом применении и отмене не установлено.
Запрещено применять Онсиор таблетки беременным и лактирующим животным, щенкам моложе 3-месячного и котятам моложе 4-месячного возраста.
Следует избегать нарушения рекомендуемого интервала между введениями препарата, так как это может привести к снижению терапевтической эффективности. В случае пропуска повторного введения препарат следует ввести как можно скорее в предусмотренной дозе.
При применении Онсиора таблеток в соответствии с настоящей инструкцией побочных явлений и осложнений у животных, как правило, не наблюдается. У кошек и собак возможны желудочно-кишечные расстройства (рвота, мягкий кал или диарея), указанные симптомы проходят самопроизвольно и не требуют применения лекарственных средств. При индивидуальной повышенной чувствительности животного к препарату и появлении аллергических реакций, применение препарата прекращают и животному назначают антигистаминные и симптоматические средства.
Онсиор таблетки не следует применять одновременно с другими нестероидными противовоспалительными препаратами и в течение 24 часов после их отмены, диуретиками, а также ингибиторами АПФ и другими лекарственными средствами с высокой степенью связывания с белками сыворотки крови из-за вероятности взаимного усиления токсического действия.
Лекарственный препарат не предназначен для применения продуктивным животным.
Меры личной профилактики:
Людям с гиперчувствительностью к компонентам препарата следует избегать прямого контакта с Онсиором таблетками. При случайном контакте лекарственного препарата с кожей или слизистыми оболочками, их необходимо немедленно промыть большим количеством воды.
Запрещается использование пустой тары из-под препарата для бытовых целей; она подлежит утилизации с бытовыми отходами.
При применении Онсиора таблеток следует соблюдать общие правила личной гигиены и техники безопасности, предусмотренные при работе с лекарственными препаратами. Во время работы запрещается курить, пить и принимать пищу. По окончании работы следует тщательно вымыть с мылом лицо и руки.
В случае появления аллергических реакций или при случайном попадании препарата в организм человека следует немедленно обратиться в медицинское учреждение (при себе иметь инструкцию по применению препарата или этикетку).
Перед применением рекомендуется проконсультироваться с ветеринарным врачом.
Страна-производитель: Франция.
Информация о составе и ингредиентах, цветовом решении, технических и иных характеристиках, стране-изготовителе, внешнем виде и комплекте поставки товара носит справочный характер и основана на последних доступных к моменту публикации сведениях. Доставка данного товара осуществляется во все регионы РФ.
Нестероидный противовоспалительный препарат для лечения воспалительных и болевых синдромов различного происхождения, в том числе являющихся последствиями хирургического вмешательства, у собак и кошек.</br>
</br>
__Действующее вещество:__ робенакоксиб.</br>
</br>
__Применение:__ Онсиор™ назначают собакам и кошкам в качестве противовоспалительного и болеутоляющего препарата при воспалительных и болевых синдромах различного происхождения, включая острые и хронические заболевания опорно-двигательного аппарата (артриты, артрозы, синовиты, вывихи), воспалительные заболевания мягких тканей, а также в качестве анальгезирующего и противовоспалительного препарата в послеоперационный период.</br>
</br>
__Срок годности и условия хранения:__ таблетки — 4 года с даты производства при температуре от 0°С до 25°С. Инъекции – 3 года с даты производства при температуре от 2°С до 8°С; после первого вскрытия флакона – 28 дней.